Airborne transmission of pathogens
Table of Contents
- Table of Contents
- This page
- General reference works about the airborne transmission of pathogens
- Review articles that deal with the airborne transmission of specific pathogens
- Studies that detected pathogens or their genetic material in air samples or air filters or in places unlikely to be touched
- Studies showing that a pathogen can infect over a distance (i.e. where animals infected each other by airborne spread)
- Animal model studies. These either infect animals by air, isolate pathogen out of animal breath, or show that animals can infect each other through the air
- Review articles and reports of airborne spread between or in buildings
- All other reports of the airborne transmission of pathogens, sorted by pathogen
- chickenpox
- coronaviruses, animal coronaviruses
- coronaviruses, SARS and MERS
- coronaviruses, SARS-COV-2/COVID-19
- Filoviruses (Marburgvirus, Ebolavirus, Restonvirus, etc.)
- Hantaviruses
- Influenza
- found influenza virus in the air - see the earlier section
- found virus or its genetic material in the air or in places unlikely to be touched
- Reports of outbreaks that occurred despite using contact precautions
- virus remained viable in air over a significant period of time
- human challenge trials or human to animal challenge trials
- Kennel cough
- lassavirus air
- lentivirus (HIV, etc) air
- leprosy (bacteria) air
- MRSA (bacteria)
- measles
- norovirus
- rabies air
- RSV and B pertussis (bacteria) air
- rhinovirus air
- tuberculosis
- Reports of true long-range airborne spread (i.e. over large distances or atmospheric)
- Quantifying virus exhaled from the breath
- Miscellaneous other topics
This page
This page lives at https://its-airborne.org/airborne-reference-works.
General reference works about the airborne transmission of pathogens
Books
List of books and review articles about airborne transmission of pathogens. Theses lists started, and for the most part still exist, as posts on Twitter and Mastodon, which can be found at the following pages. They may not be as up-to-date as this page, however.
- Books:
- Review articles:
- Moulton, Forest Ray, ed. 1942. Aerobiology. American Association for the Advancement of Science. https://www.google.ca/books/edition/Aerobiology/DNdfAAAAMAAJ
- William Firth Wells. 1955. Airborne Contagion and Air Hygiene: An Ecological Study of Droplet Infections https://archive.org/details/airborne-contagion and https://books.google.ca/books/about/Airborne_Contagion_and_Air_Hygiene.html?id=T8nVAAAAMAAJ
- I happily present to you, kind readers, the Bible of airborne transmission of pathogens. It is on Internet Archive but also on Google Books. Click the gear icon to download it as a PDF. Interesting that I found it on Google Books, because I thought I had searched for this book there in the past and it wasn’t available for download.
- Richard L. Riley, Francis O’Grady. 1961. Airborne Infection: Transmission and Control. https://books.google.ca/books?id=qztrAAAAMAAJ
- Riley worked with Mr. Wells on aerobiology his whole life.
- Mr. Wells wrote the book I mentioned a few posts up. He was the husband of Dr. Wells, who also worked on aerobiology but passed away in the 40s. WHO. 2009. “Natural ventilation in health care for infection control”
- A World Health Organization #WHO publication, which they never mention for some odd reason. https://www.ncbi.nlm.nih.gov/books/NBK143284/.
- Kundsin, Ruth B, editor. 1980. Airborne Contagion. New York, N.Y. : New York Academy of Sciences. http://archive.org/details/airbornecontagio0000unse.
- Papers include:
- “Spread of TB via recirculated air on a naval vessel”,
- “The role of ventilation in the spread of measles in an elementary school”, and
- Langmuir, former CDC director, admitting he got airborne spread wrong but also herd immunity. What is wrong with this field, honestly. This piecs is, interestingly, not in the Langmuir collection https://hollisarchives.lib.harvard.edu/repositories/14/resources/4634/collection_organization#tree::archival_object_1289931. Hmm.
- From “Airborne Contagion”:
-
“Airborne transmission is the most important mode of transmission of respiratory infections from person to person indoors. It may well be the most important mode of transmission for other human infections not considered as primarily respiratory. There is published evidence of droplet nuclei transmission of hepatitis B virus, smallpox, rabies, chicken pox, mumps, measles as well as tuberculosis. I am deeply grateful to Dr. Lloyd G. Herman, Dr. Richard L. Riley, and Dr. Carl W. Walter for their interest, support, and total dedication to the theme of this conference: airborne contagion.”
-
- The titles of the various articles in Kundsin’s conference compilation:
-
Part I. History and Epidemiology
-
Historical Background. By RICHARD L. RILEY 3
-
Spread of Tuberculosis via Recirculated Air in a Naval Vessel: The Byrd Study. By VERNON N. HOUK 10
-
The Role of Ventilation in the Spread of Measles in an Elementary School. By EDWARD C. RILEY 25
-
Changing Concepts of Airborne Infection of Acute Contagious Diseases: A Reconsideration of Classic Epidemiologic Theories. By ALEXANDER D. LANGMUIR 35
-
Part II. Epidemiology
-
The Epidemiology of Influenza in Humans. By MICHAEL B. GREGG 45
-
Epidemiology of the Common Cold. By JACK M. GWALTNEY, JR 54
-
Legionellosis: Evidence of Airborne Transmission. By DAVID W. FRASER 61
-
Legionellosis: Environmental Aspects. By G. F. MALLISON 67
-
Physics of Airborne Particles and Their Deposition in the Lung. By PAUL E. MORROW 71
-
A Tribute to William Firth Wells. By EDWARD C. RILEY 81
-
Part III. Bacteria as Agents of Airborne Contagion
-
Inhalation Anthrax. By PHILIP S. BRACHMAN 83
-
Aerosol Dissemination of Bacterial Plant Pathogens. By MONTY D. HARRISON 94
-
Airborne Spread of Brucellosis. By ARNOLD F. KAUFMANN, MARSHALL D. Fox, JOHN M. BOYCE, DANIEL C. ANDERSON, MORRIS E. POTTER, WILLIAM J. MARTONE, and CHARLOTTE M. PATTON 105
-
Part IV. Fungi as Agents of Airborne Contagion
-
Introduction. By LLOYD G. HERMAN 115
-
Aerial Dissemination of Fungal Spores. By DONALD E. AYLOR and PAUL E. WAGGONER 116
-
(Philosophical) Review of Air Currents as a Continuing Vector. By CHARLOTTE C. CAMPBELL 123
-
Aspergillus in Patient Care Areas. By LLOYD G. HERMAN 140
-
Part V. Viruses as Agents of Airborne Contagion
-
Viruses as Agents of Airborne Contagion. By VERNON KNIGHT 147
-
Aerosol Spread of Plant Viruses: Potential Role in Disease Outbreaks. By ERNEST E. BANTTARI AND JAMES R. VENETTE 167
-
Overview of Airborne Contagion in Animals. By LAWRENCE A. FALK, JR. and RONALD D. HUNT 174
-
Spread of Plant Viruses and Spiroplasmas through Airborne Vectors. By KARL MARAMOROSCH 179
-
Part VI. Airborne Transmission—Other Considerations
-
Long-Range Transmission of Bacteria. By AKE BOVALLIUS, ROGER ROFFEY, and EVA HENNINGSON 186
-
Surf-to-Wind Transfer of Viruses. By EDWARD R. BAYLOR and MARTHA B. BAYLOR 201
-
Spread of Microorganisms by Air-Conditioning Systems—Especially in Hospitals. By K. O. GUNDERMANN 209
-
The Role of Airborne Bacteria in the Contamination of Fine Particle Nebulizers and the Development of Noscomial Pneumonia. By STEVEN G. KELSEN and MARYANNE MCGUCKIN 218
-
Air Sampling in Hospitals. By DIETER H. M. GROSCHEL 230
-
Techniques Used for Sampling Airborne Microorganisms Associated with Industrial Clean Rooms and Spacecraft Assembly Areas. By MARTIN
-
S. FAVERO and JOHN R. PULEO 241
-
Part VII. Airborne Infections in Hospitals
-
Documentation of Airborne Infection During Surgery. By RUTH B. KUNDSIN 255
-
Reduction of Deep Sepsis Following Total Hip Arthroplasty. By ROBERT H. FITZGERALD, JR 262
-
Ultraviolet Light for the Control of Airborne Bacteria in the Operating Room. By J. LEONARD GOLDNER, MARY MOGGIO, STEPHEN F. BEISSINGER, and DONALD E. MCCOLLUM 271
-
Ultraviolet Radiation and Reduction of Deep Wound Infection Following Hip and Knee Arthroplasty. By J. DRENNAN LOWELL, RUTH B. KUNDSIN, CHARLES M. SCHWARTZ, and DEBORAH POZIN 285
-
The Treatment of Burn Patients in a Laminar Airflow Environment. By ROBERT H. DEMLING and JEANNE MALY 294
-
The Contribution of A Bacterially Isolated Environment to the Prevention of Infections in Seriously Burned Patients. By GENN E. BEHRINGER and JOHN F. BURKE 300
-
Part VIII. Prevention and Control
-
Speculations on the Possible Effects of the Indoor Air on Airborne Contagion. By DONALD F. PROCTOR 308
-
Prevention and Control of Airborne Infection in Hospitals. By CARL W. WALTER 312
-
Prevention and Control of Airborne Infection in the Community. By RICHARD L. RILEY 331
-
Index of Contributors 341
-
Financial assistance was received from:
-
• BOEHRINGER INGELHEIM LTD.
-
• JOHNSON & JOHNSON PRODUCTS, INC.
-
• MERCK SHARP & DOHME RESEARCH LABORATORIES
-
• A. H. ROBBINS COMPANY
-
• NATIONAL INSTITUTE OF ALLERGY AND INFECTIOUS DISEASES—
-
FOGARTY INTERNATIONAL CENTER
-
• OFFICE OF NAVAL RESEARCH
-
- Papers include:
- Maureen E. Lacey and Jonathan S. West. 2006. _The Air Spora+. 2006 : 15–34. The Aerobiology Pathway.
- doi:10.1007/978-0-387-30253-9_2
- PMCID: PMC7120664
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7120664/
- Salem, Harry, and Sidney A Katz, eds. - Aerobiology The Toxicology of Airborne Pathogens. 2016. Cambridge: Royal Society of Chemistry, 2016. https://doi.org/10.1039/9781849737913-FP007
Review articles about airborne transmission of pathogens
This is not all of the reviews, just the top ones. These reviews are all (mostly) written by people who work on aerosols. They are broad summaries (hence, “reviews”) of the field. Start here with your reading. You can then drill down to the actual studies if you want. That means they aren’t public health people fooling around with an air sampler they just bought and unboxed. Not joking.
- 1987 Sattar, Syed A., Mohammad Khalid Ijaz, and Charles P. Gerba. ‘Spread of Viral Infections by Aerosols’. Critical Reviews in Env Control 17, no. 2 (January 1987): 89–131. https://doi.org/10.1080/10643388709388331
- So as not to mislead you into thinking this is a recent development, I will just provide ONE reference from 1987.
- These people were never confused about 5um, etc.
-
Aerosols are dispersions in air of particles of a variety of sizes. The larger of these particles rapidly settle out, but particles of smaller size can remain suspended in air for longer periods. If the air were to be perfectly still, it would take a 100 ym diameter particle 10 sec to fall through the height of an ordinary room (about 3 m); particles with diameters of 40, 20, and 10 um would require 1, 4, and 17 min, respectively, to settle out under the same set of conditions.?? Under real conditions, the time during which aerosol particles remain suspended and the distance which they can travel from the point of their generation are greatly influenced by airflow and turbulance. Many common and natural activities in the domestic, work, or animal husbandry environments regularly result in the generation of aerosols from microbially contami- nated liquids or the resuspension in air of previously dried infectious material. For example, sneezing, coughing, and even speaking by persons carrying viruses in their mouth and respiratory tract frequently lead to the aerosolization of viruses.*$ The particles produced during sneezing and speaking (particularly when pronouncing sibi- lants) are generally larger and most of them rapidly settle out of air. Coughing, on the other hand, is known to produce more small-particle aerosols which are potentially better suited for the airborne spread of viral infections.®
- Oh, re: the lab leak issue, enjoy last line.
- Again, from 1987.
-
VI. VIRAL AEROSOLS GENERATED IN THE WORK AND HOME ENVIRONMENT The first record of a laboratory-acquired infection dates back to 1885.!! Since then, much attention has been given to the possible spread of viruses and other infectious agents to laboratory workers involved in their handling. Sulkin and Pike’** summa- rized the data from 222 reports of laboratory-acquired infections. The following types of viruses were among the infectious agents involved: eastern, western, and Venezuelan equine encephalitis; Russian spring summer encephalitis; louping ill; lymphocytic cho- riomeningitis; poliomyelitis; encephalomyocarditis; Newcastle disease; yellow fever; dengue fever; Rift Valley fever; Colorado tick fever; mumps; influenza; hepatitis; ru- bella; and agents of viral diarrhea. Analysis of the reports indicated that 30% of the infections were due to contaminated laboratory air.
-
- And exposure to air leading to infection, and filtration slashing infection. It’s all there, if you are open to it.
-
Marek’s disease Chickens Exposure to effluent air from ““donor cages’ housing 164 virus (MDV) infected animals resulted in a high incidence of MDV infection in test chicks; passage of contaminated air through certain filters partially or completely pre- vented such infection
-
- Same article, from 1987:
-
E. Lymphocytic Choriomeningitis An outbreak of lymphocytic choriomeningitis (LCM) occurred in 1972 to 1973 in personnel at a medical center in Rochester, New York.’*® Epidemiological and virol- ogical studies indicated that the source of the infection were Syrian hamsters being used there for tumor research. Cell cultures derived from these animals were also found to be contaminated with the virus. The cases of human infection were shown to occur not only through direct contact with the animals, but also from mere presence in the room where the animals were being held.
-
- and
-
… studies indicated that the source of the infection were Syrian hamsters being used there for tumor research. Cell cultures derived from these animals were also found …
-
- Morawska, L. “Droplet Fate in Indoor Environments, or Can We Prevent the Spread of Infection?Abstract.” Indoor Air 16, no. 5 (2006): 335–47. https://doi.org/10.1111/j.1600-0668.2006.00432.x.
- 2006 Tang, J. W., Y. Li, I. Eames, P. K. S. Chan, and G. L. Ridgway. ‘Factors Involved in the Aerosol Transmission of Infection and Control of Ventilation in Healthcare Premises’. The Journal of Hospital Infection 64, no. 2 (October 2006): 100–114. https://doi.org/10.1016/j.jhin.2006.05.022 or https://www.journalofhospitalinfection.com/article/S0195-6701(06)00286-6/fulltext.
- 2011 By mechanical engineers in Canada. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3226423/.
- 2016-09. Ijaz, M. Khalid et al and Sattar, Syed A.. ‘Generic Aspects of the Airborne Spread of Human Pathogens Indoors and Emerging Air Decontamination Technologies’. American Journal of Infection Control 44, no. 9 (September 2016): S109–20. https://doi.org/10.1016/j.ajic.2016.06.008.
- Same authors as in 1980s, still writing about aerosol.
- 2019 Tellier, Raymond, Yuguo Li, Benjamin J. Cowling, and Julian W. Tang. ‘Recognition of Aerosol Transmission of Infectious Agents: A Commentary’. BMC Infectious Diseases 19, no. 1 (31 January 2019): 101. https://doi.org/10.1186/s12879-019-3707-y.
- Top people
- 2020 March. Bourouiba, Lydia. ‘Turbulent Gas Clouds and Respiratory Pathogen Emissions: Potential Implications for Reducing Transmission of COVID-19’. JAMA, 26 March 2020. https://jamanetwork.com/journals/jama/fullarticle/2763852. – droplets travel further than 2 meters
- 2020 April. Bahl, Prateek, Doolan, de Silva, Chughtai, Bourouiba, and MacIntyre. ‘Airborne or Droplet Precautions for Health Workers Treating Coronavirus Disease 2019?’ The Journal of Infectious Diseases, no. jiaa189 (16 April 2020). https://doi.org/10.1093/infdis/jiaa189.
- 2020 May. Anderson, Elizabeth L., Paul Turnham, John R. Griffin, and Chester C. Clarke. ‘Consideration of the Aerosol Transmission for COVID‐19 and Public Health’. Risk Analysis 40, no. 5 (May 2020): 902–7. https://doi.org/10.1111/risa.13500.
- Another saying we should look at aerosol.
- 2020 June. Morawska, Lidia, and Donald K Milton. ‘It Is Time to Address Airborne Transmission of Coronavirus Disease 2019 (COVID-19)’. Clinical Infectious Diseases, 6 July 2020, ciaa939. https://doi.org/10.1093/cid/ciaa939.
- 2020 June. Morawska, Lidia, and Junji Cao. ‘Airborne Transmission of SARS-CoV-2: The World Should Face the Reality’. Environ. Int. 139 (June 2020): 105730. https://doi.org/10.1016/j.envint.2020.105730.
- 2020-08. Wilson, N. M., A. Norton, F. P. Young, and D. W. Collins. ‘Airborne Transmission of Severe Acute Respiratory Syndrome Coronavirus-2 to Healthcare Workers: A Narrative Review’. Anaesthesia 75, no. 8 (August 2020): 1086–95. https://doi.org/10.1111/anae.15093.
- Morawska, Lidia, Julian W. Tang, William Bahnfleth, Philomena M. Bluyssen, Atze Boerstra, Giorgio Buonanno, Junji Cao, et al. 2020 Sept. ‘How Can Airborne Transmission of COVID-19 Indoors Be Minimised?’ Env Intl 142 (1 September 2020): 105832. https://doi.org/10.1016/j.envint.2020.105832.
- Prather, Kimberly A, Linsey C Marr, Robert T Schooley, Melissa A McDiarmid, Mary E Wilson, and Donald K Milton. 2020 Oct. ‘Airborne Transmission of SARS-CoV-2’. Science 370, no. 6514 (16 October 2020): 303–4. https://doi.org/10.1126/science.abf0521.
- Tang, Song, Yixin Mao, Rachael M. Jones, Qiyue Tan, John S. Ji, Na Li, Jin Shen, et al. 2020-11. ‘Aerosol Transmission of SARS-CoV-2? Evidence, Prevention and Control’. Environment International 144 (1 November 2020): 106039. https://doi.org/10.1016/j.envint.2020.106039.
- Bourouiba, Lydia. 2021. ‘Fluid Dynamics of Respiratory Infectious Diseases’. Annual Review of Biomedical Engineering 23, no. 1 (2021): 547–77. https://www.annualreviews.org/doi/10.1146/annurev-bioeng-111820-025044.
- Review from 2021, also should be in the “top” pile.
- Samet, Jonathan M, Kimberly Prather, Georges Benjamin, Seema Lakdawala, John-Martin Lowe, Arthur Reingold, John Volckens, and Linsey Marr. 2021 Jan. ‘Airborne Transmission of SARS-CoV-2: What We Know’. Clin. Infect. Dis., 18 January 2021. https://doi.org/10.1093/cid/ciab039.
- Tang, J W, Bahnfleth, Bluyssen, Buonanno, Jimenez, J Kurnitski, Y Li, et al. 2021 April. ‘Dismantling Myths on the Airborne Transmission of Severe Acute Respiratory Syndrome Coronavirus-2 (SARS-CoV-2)’. J. Hosp. Infect. 110 (April 2021): 89–96. https://doi.org/10.1016/j.jhin.2020.12.022.
- Morawska, Lidia, Joseph Allen, William Bahnfleth, Philomena M Bluyssen, Atze Boerstra, Giorgio Buonanno, Junji Cao, et al. 2021 May. ‘A Paradigm Shift to Combat Indoor Respiratory Infection’. Science 372, no. 6543 (14 May 2021): 689–91. https://doi.org/10.1126/science.abg2025.
- Greenhalgh et al. 2021 May. ‘Ten Scientific Reasons in Support of Airborne Transmission of SARS-CoV-2’. The Lancet 397, no. 10285 (1 May 2021): 1603–5. https://doi.org/10.1016/S0140-6736(21)00869-2.
- Wang et al. 2021 August. ‘Airborne Transmission of Respiratory Viruses’. Science 373, no. 6558 (27 August 2021). https://doi.org/10.1126/science.abd9149.
- Leung, Nancy H. L. 2021-08. ‘Transmissibility and Transmission of Respiratory Viruses’. Nature Reviews Microbiology 19, no. 8 (August 2021): 528–45. https://doi.org/10.1038/s41579-021-00535-6.
- Addleman, Sarah, Victor Leung, Leyla Asadi, Abdu Sharkawy, and Jennifer McDonald. ‘Mitigating Airborne Transmission of SARS-CoV-2’. CMAJ, 1 January 2021. https://doi.org/10.1503/cmaj.210830.
- Randomly found another review, discussing SARS and MERS airborne, amongst others. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7150194/#Art1.
- Editorial in Indoor Air, entitled “all respiratory viruses are airborne.” https://onlinelibrary.wiley.com/doi/10.1111/ina.12937.
- Tang, Tellier and Yuguo Li are all experts.
Historical review papers about the airborne transmission of pathogens
- Jimenez, Jose L., Linsey C. Marr, Katherine Randall, et al. “What Were the Historical Reasons for the Resistance to Recognizing Airborne Transmission during the COVID-19 Pandemic?” Indoor Air 32, no. 8 (2022): e13070. https://doi.org/10.1111/ina.13070.
- for a more comprehensive historical overview, this article spearheaded by jljcolorado is very good, I dare say. Full disclosure: I am a co-author.
- Randall, Ewing, Marr, Jimenez and Bourouiba. Nov 2021. ‘How Did We Get Here: What Are Droplets and Aerosols and How Far Do They Go? A Historical Perspective on the Transmission of Respiratory Infectious Diseases’. Interface Focus 11, no. 6 (Nov 2021). https://doi.org/10.1098/rsfs.2021.0049
- Pepper, Ian L., and Charles P. Gerba. 2015. ‘Aeromicrobiology’. Environmental Microbiology, 89–110. https://doi.org/10.1016/B978-0-12-394626-3.00005-3
- But very disappointed they repeated the 5uM fiction, and the idea of droplet. So that’s a big negative. And Brankston (pic 4) is garbage. Otherwise, the chapter is a good overview of the area.
- Information Box 5.3: examples of airborne plant pathogens (and then a list of about 16 pathogens)
- Information Box 5.4: Examples of airborne animal pathogens. And then a list of about 17 items.
-
A table of aerosolized endotoxin concentrations detected downwind of biosolids operations (waste treatment facilities)
-
Influenza virus transmission among humans can occur via four mechanisms: by direct contact with infected indi- viduals: by indirect contact with contaminated objects of fomites; by inhalation of droplets that contain the virus; or by inhalation of aerosolized virus. Interestingly. despitc 70 years of research since the influenza A virus was dis- covered, ther is sill debate about the modes of influenza transmission, specifically whether influenza is mainly transmitted via true bioaerosols, or by droplets, or by direct or indirect contact (Brankston ef al 2007).
- 2022 Moreno T and Gibbons W. ‘Aerosol Transmission of Human Pathogens: From Miasmata to Modern Viral Pandemics and Their Preservation Potential in the Anthropocene Record’. Geoscience Frontiers 13, no. 6 (November 2022): 101282. https://doi.org/10.1016/j.gsf.2021.101282
Review articles that deal with the airborne transmission of specific pathogens
coronaviruses, generally
- Pozzi, Paolo, Alessio Soggiu, Luigi Bonizzi, Nati Elkin, and Alfonso Zecconi. “Airborne Coronaviruses: Observations from Veterinary Experience.” Pathogens 10, no. 5 (2021): 628. https://doi.org/10.3390/pathogens10050628.
“Porcine respiratory CoV in swine (PRCV) is known for its aerogenic spread between herds. The virus is shed from nasal secretions for less than 2 weeks, and diffuses through droplets and aerosol; nasal shedding of PRCV in experimentally infected pigs occurs through postexposure until 10 days.” [Pozzi et al., 2021, p. 2] “Population density plays a major role in the spread of PRCV. Indeed, in areas of high swine population density, PRCV can spread several kilometers and the risk of a farm to become infected increases as neighboring herds increase in size within a radius from 2–10 km.” [Pozzi et al., 2021, p. 3] “Porcine epidemic diarrhea virus (PEDV), another member of CoVs in pigs, is capable of aerogenic spread; it was detected in aerosols of different sizes and in concentrations ranging from 1.3 × 106 (in particles of 0.4–0.7 μm) to 3.5 × 108 RNA copies/m3 (in particles of 9.0–10.0 μm), following experimental infection [20]. In an experimental study, PEDV was detected as airborne for 24 days post-infection, and the virus remained viable and able to re-infect other pigs [20]. Concerning airborne diffusion, higher quantities of PEDV were found associated with larger particles. Indeed, under field conditions, PEDV viral RNA was identified at significantly higher concentrations in particles >3.3 μm in diameter compared with those of 0.4–1.1 μm. The infectious particles with a size <10 μm may increase the risk of serious health implications because they are able to penetrate the lower respiratory tract to establish infection. PEDV showed a statistically higher concentration in large-size particles (>9 μm) vs. particles ranging from 1.1 μm to 5.8–9.0 μm [15]. The aerosols exhaled from human patients with respiratory infections have shown a predominance of different pathogens, both bacteria and viruses, in small-size particles (<5 μm) [21].” [Pozzi et al., 2021, p. 3] “Bovine CoV (BCoV), another CoV causing respiratory disease and diarrhea in calves and winter dysentery in adult cattle, considered endemic in cattle populations, is known for its nasal shedding, further than fecal, along 1 to 11–12 days post-infection” [Pozzi et al., 2021, p. 3] “the aerosol/airborne transmission route was also confirmed” [Pozzi et al., 2021, p. 3] “Infected calves were demonstrated shedding BCoV for up to 13 days, with the rapid spread of BCV to all susceptible animals within a unit.” [Pozzi et al., 2021, p. 3] “Recent outbreaks of SARS-CoV-2/COVID-19 in fur-mink farms confirmed both the airborne transmission of the virus” [Pozzi et al., 2021, p. 3]
Coronavirus, IBV (a chicken coronavirus)
- (Not sampling, just a review article.)
- “The virus spreads by both air and the fecal-oral route”

Influenza
- Tellier. 2006. Review of Aerosol Transmission of Influenza A Virus.
- refers to a number of studies on page 1658, left column

Studies that detected pathogens or their genetic material in air samples or air filters or in places unlikely to be touched
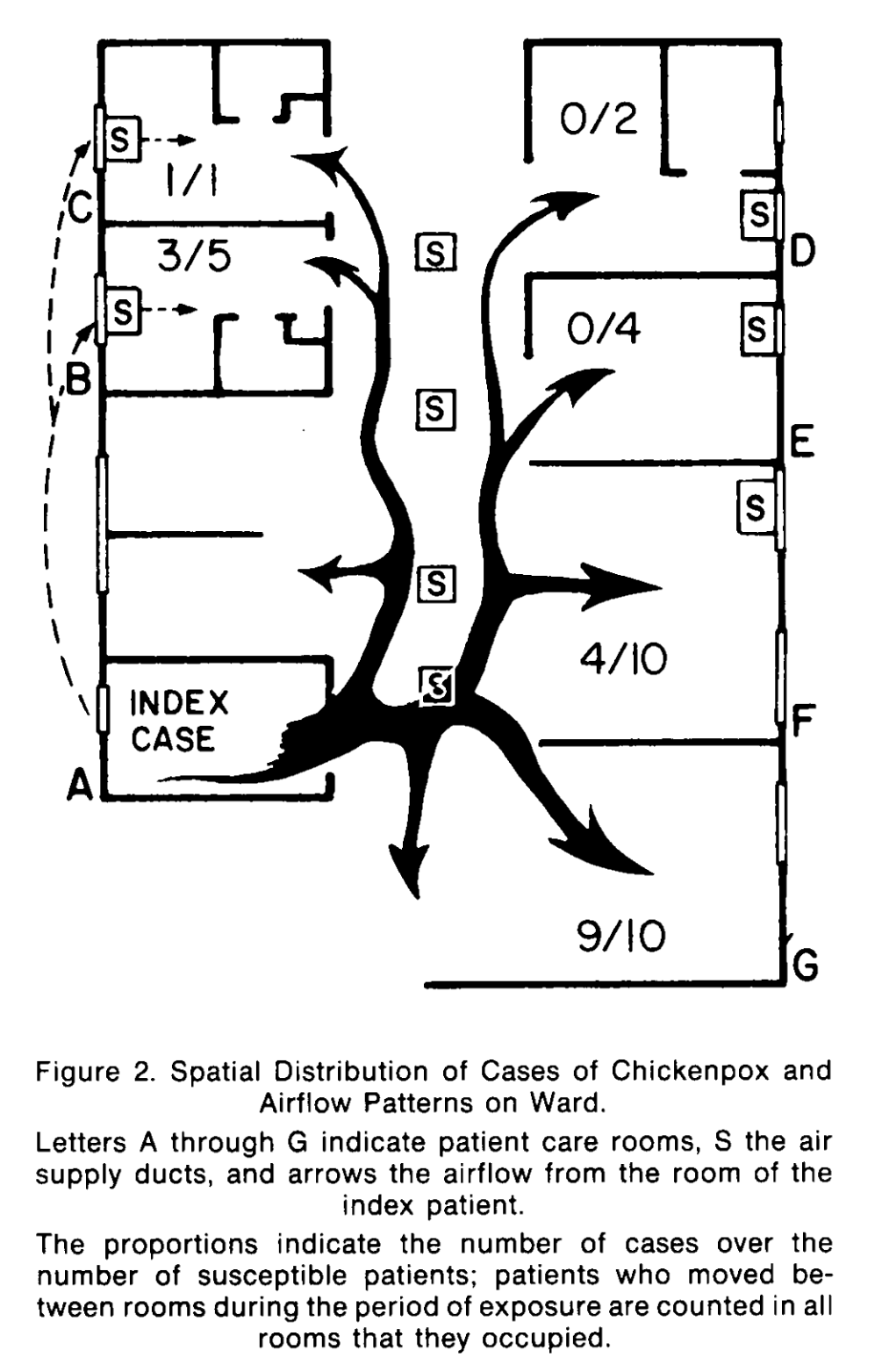
Chickenpox
- 1994-01. Sawyer, M. H., C. J. Chamberlin, Y. N. Wu, N. Aintablian, and M. R. Wallace. ‘Detection of Varicella-Zoster Virus DNA in Air Samples from Hospital Rooms’. The Journal of Infectious Diseases 169, no. 1 (January 1994): 91–94. https://doi.org/10.1093/infdis/169.1.91
- “VZV was detected 1.2-5.5 m from patients’ beds and for 1-6 days following onset of rash. On some occasions, VZV DNA could be detected outside the hospital isolation rooms housing patients.”
- my notes:
- The biggest attack rate was in room G, 9/10 people caught it - the HVAC was broken in that room.
- Room C was under strict gown and gloves precautions and patient still caught it.
- This is because chickenpox, like most viruses, predominantly transmits by air.
- The rash is a red herring. Rash does not indicate the pathogen was caught by touch. If the pathogen is throughout your body, it’s in your blood and systemically disseminated. Also in saliva.
- Brankston, a very anti-airborne article, cited Sawyer as having proven chickenpox was airborne. Brankston was arguing that if influenza was also airborne, we’d see articles about it like Sawyer for chickenpox. It’s a specious argument but that’s what was said.
-
“If influenza could be transmitted over long distances via the airborne route, then we would have expected to review studies citing evidence similar to that reported for the varicella zoster virus and Mycobacterium tuberculosis, both of which undergo known airborne transmission.” [then citing Sawyer, above, that varicella DNA being found in the air 1.2-5.5m from beds up to 24 h later]
- Brankston also said
-
“Two epidemiological studies of varicella transmission were supplemented by airflow studies that showed directional airflow consistent with the spread of the illness.50,51”
-
- and cited these two other famous chickenpox articles:
- [50] Gustafson et al An outbreak of airborne nosocomial varicella. Pediatrics 1982; 70: 550–56. 51
- [51] Leclair et al Airborne transmission of chickenpox in a hospital. N Engl J Med 1980; 302: 450–53.
- [50] Gustafson described the following:
- Index remained in isolation in room. 8 of 70 caught it, and seems like on a quick skim all 8 were there one afternoon.
- Hmm, hit rate far less than some case descriptions of COVID-19. One wonders if people are reading these old case descriptions or not.
- [51] Leclair was a report of a chickenpox patient on a ward who infected 15 others
- The introduction says chickenpox precautions are airborne, but “airborne spread has never been unequivocally demonstrated” and “Moreover, the low secondary attack rate after exposure to chicken pox in nonresidential settings suggests that airborne spread is less important than …contact.”
- “We describe an epidemic of chickenpox occurring in a hospital in which airflow studies [prove] airborne.”
-
- Asano 1999. https://pubmed.ncbi.nlm.nih.gov/10224205/
- not found in the air, but found in the AC filter and multiple surfaces in a house
- Yoshikawa 2001. https://pubmed.ncbi.nlm.nih.gov/11130889/
- Suzuki 2002. https://pubmed.ncbi.nlm.nih.gov/11857538/
- Suzuki 2003. https://pubmed.ncbi.nlm.nih.gov/12911484/
- not found in the air, but found in air purifier filters in the houses of infected children
- Suzuki 2004. https://pubmed.ncbi.nlm.nih.gov/12911484/
Coronavirus, SARS-CoV-2
Coronavirus, SARS-CoV-2 - studies that found viable pathogen in air samples
- 2020-07. Santarpia et al. ‘Aerosol and Surface Contamination of SARS-CoV-2 Observed in Quarantine and Isolation Care’ Sci Rep 10, no 1 (29 July 2020): 12732. https://www.nature.com/articles/s41598-020-69286-3
- Santarpia originally reported recovering RNA from the air only, but later the paper was amended because they managed to culture the virus from the samples.
- 2020-11. Lednicky, John A. et al. ‘Viable SARS-CoV-2 in the Air of a Hospital Room with COVID-19 Patients’. Intl J Inf Dis 100 (1 Nov 2020): 476–82. https://www.ijidonline.com/article/S1201-9712(20)30739-6/fulltext
- Viable SARS-CoV-2 virus recovered from a patient room in a hospital in the United States.
- 2020-11. Razzini, Katia et al. ‘SARS-CoV-2 RNA Detection in the Air and on Surfaces in the COVID-19 Ward of a Hospital in Milan, Italy’. The Science of the Total Environment 742 (10 November 2020): 140540. https://www.sciencedirect.com/science/article/pii/S0048969720340626?via%3Dihub
- SARS-CoV-2 RNA in hospital air, Italy. November 2020
- Adenaiye. 2021. https://academic.oup.com/cid/advance-article/doi/10.1093/cid/ciab797/6370149
- viable virus isolated from aerosol samples in a laboratory setting.
- 2021-07. Lednicky et al., “Isolation of SARS-CoV-2 from the Air in a Car Driven by a COVID Patient with Mild Illness.” x
- 2021-08. Santarpia, Joshua L., Vicki L. Herrera, Danielle N. Rivera, et al. “The Size and Culturability of Patient-Generated SARS-CoV-2 Aerosol.” Journal of Exposure Science & Environmental Epidemiology 32, no. 5 (2022): 706–11. https://doi.org/10.1038/s41370-021-00376-8. https://www.nature.com/articles/s41370-021-00376-8
- 2021-11. Kotwa et al. ‘Surface and Air Contamination with SARS-CoV-2 from Hospitalized COVID-19 Patients in Toronto, Canada, March-May 2020’. J Inf Dis, 27 November 2021, jiab578.
- The conclusion of the following paper is a defensive “it’s safe for workers” article. They say b/c “ infrequent recovery of infectious virus suggests risk is limited” (they took 36 samples got 6 cultured not bad I bet, actually). However, they were able to sample live virus from air
- https://academic.oup.com/jid/article/225/5/768/6444802
- 2022-01. Ang, Alicia Xy, Irvan Luhung, Bintou A. Ahidjo, et al. “Airborne SARS-CoV-2 Surveillance in Hospital Environment Using High-Flowrate Air Samplers and Its Comparison to Surface Sampling.” Indoor Air 32, no. 1 (2022): e12930. https://doi.org/10.1111/ina.12930.
- sampled air in hospital. Found SARS-CoV-2 RNA in the air. Could not culture it, but that might have been due to the high flow rate needed to successfully capture virus in a hospital, where the air changes were already quite high. Found positive samples in a sampler 5.5 metres from the patient.
- 2023-01. Kitagawa, Hiroki, Toshihito Nomura, Yuki Kaiki, et al. “Viable SARS-CoV-2 Detected in the Air of Hospital Rooms of Patients with COVID-19 with an Early Infection.” International Journal of Infectious Diseases: IJID: Official Publication of the International Society for Infectious Diseases 126 (January 2023): 73–78. https://doi.org/10.1016/j.ijid.2022.11.003.
- 2023-03. Fortin et al ‘Detection of Viable SARS-CoV-2 in Retrospective Analysis of Aerosol Samples Collected from Hospital Rooms of Patients with COVID-19’. Clin Microb Inf (22 March 2023). - https://www.clinicalmicrobiologyandinfection.com/article/S1198-743X(23)00135-0/fulltext
- Viable SARS-CoV-2 from hospital rooms. March 2023
Coronavirus, SARS-CoV-2 - studies that found RNA in air samples
- 2020-04-23 Park et al.
- While I am side tracked by epidemiological numbers, including attack rates, I point out that by April 2020, we had a Korean call centre experience an almost 46% attack rate for SARS-CoV-2
- 97 ppl infected, 94 were on 11th floor call centre of 216 people. 89 sympt, 4 presymp, 4 asymptomatic over 14 d quarantine. Secondary attack rate of 46%. This should have been ID doctors’ wake up call that this was airborne, but no, the denial was strong in this group.
- 2020-04. Liu, Yuan, Zhi Ning, Yu Chen, Ming Guo, Yingle Liu, Nirmal Kumar Gali, Li Sun, et al. ‘Aerodynamic Analysis of SARS-CoV-2 in Two Wuhan Hospitals’. Nature 582, no. 7813 (April 2020): 557–60. https://www.nature.com/articles/s41586-020-2271-3
- Found SARS-CoV-2 RNA in hospital air, China
- 2020-05. Chia, Po Ying, Kristen Kelli Coleman, et al. ‘Detection of Air and Surface Contamination by SARS-CoV-2 in Hospital Rooms of Infected Patients’. Nature Communications 11, no. 1 (29 May 2020): 2800. https://www.nature.com/articles/s41467-020-16670-2
- Found SARS-CoV-2 RNA in hospital air in Singapore
- 2020-11. Nissen, Karolina et al. ‘Long-Distance Airborne Dispersal of SARS-CoV-2 in COVID-19 Wards’. Scientific Reports 10, no. 1 (11 November 2020): 19589. https://www.nature.com/articles/s41598-020-76442-2
- SARS-CoV-2 RNA in Swedish hospital ventilation system.
- Over long range.
- 2021-01. López, Jorge Hernández, Álvaro Santos Romo, Daniel Coronado Molina, et al. “Detection of Sars-Cov-2 in the Air of Two Hospitals in Hermosillo, Sonora, México, Utilizing a Low-Cost Environmental Monitoring System.” International Journal of Infectious Diseases: IJID: Official Publication of the International Society for Infectious Diseases 102 (January 2021): 478–82. https://doi.org/10.1016/j.ijid.2020.10.089.
- “The current results are in accordance with those published about the search for SARS-CoV-2 in the environmental air, showing that the virus can be found in the air in the hospital zones where COVID-19 patients are placed (Faridi et al., 2020; Wang et al., 2020a, 2020b; Wu et al., 2020a, 2020b).” (López et al., 2021, p. 481)
- 2021-02. Moore et al. ‘Detection of SARS-CoV-2 within the Healthcare Environment: A Multi-Centre Study Conducted during the First Wave of the COVID-19 Outbreak in England’. J Hosp Inf 108 (1 February 2021): 189–96. https://www.journalofhospitalinfection.com/article/S0195-6701(20)30548-X/fulltext
- These authors were mostly sampling surfaces, but they found SARS-CoV-2 RNA in air. Couldn’t culture live virus from surface samples. I guess that means no touch right? 🤡🚗
- 2021-06. Horve, Patrick F. et al. ‘Identification of SARS-CoV-2 RNA in Healthcare Heating, Ventilation, and Air Conditioning Units’. Indoor Air, 29 June 2021
https://onlinelibrary.wiley.com/doi/10.1111/ina.12898
- SARS-CoV-2 RNA found in air vents.
- 2021-11. Man, P. de, et al. ‘Airborne SARS-CoV-2 in Home and Hospital Environments Investigated with a High-Powered Air Sampler’. J Hosp Inf 119 (1 January 2022): 126–31. https://www.journalofhospitalinfection.com/article/S0195-6701(21)00382-0/fulltext x
- High volume sampler used to find SARS-CoV-2 RNA in homes and hospitals.
- 2022-03. Rufino de Sousa, Nuno, Laura Steponaviciute, Lucille Margerie, et al. “Detection and Isolation of Airborne SARS-CoV-2 in a Hospital Setting.” Indoor Air 32, no. 3 (2022): e13023. https://doi.org/10.1111/ina.13023.
-
“To this end, we employed a novel electrostatic collector to sample air from rooms occupied by COVID-19 patients in a major Swedish hospital. Electrostatic air sampling in conjunction with extraction-free, reverse-transcriptase polymerase chain reaction (hid-RT-PCR) enabled detection of SARS-CoV-2 in air from patient rooms (9/22; 41%) and adjoining anterooms (10/22; 45%). Detection with hidRT-PCR was concomitant with viral RNA presence on the surface of exhaust ventilation channels in patients and anterooms more than 2 m from the COVID-19 patient. Importantly, it was possible to detect active SARS-CoV-2 particles from room air, with a total of 496 plaque-forming units (PFUs) being isolated, establishing the presence of infectious, airborne SARS-CoV-2 in rooms occupied by COVID-19 patients. Our results support circulation of SARS-CoV-2 via aerosols and urge the revision of existing infection control frameworks to include airborne transmission.” (Rufino de Sousa et al., 2022, p. 1)
-
- 2022-04. Buonanno, G., A. Robotto, E. Brizio, L. Morawska, A. Civra, F. Corino, D. Lembo, G. Ficco, and L. Stabile. ‘Link between SARS-CoV-2 Emissions and Airborne Concentrations: Closing the Gap in Understanding’. Journal of Hazardous Materials 428 (15 April 2022): 128279. https://doi.org/10.1016/j.jhazmat.2022.128279
- Tested viral load of patients as against virus in the air around those patients.
- 2022 Kurver et al. SARS-CoV-2 RNA in exhaled air of hospitalized COVID-19 patients. https://www.nature.com/articles/s41598-022-13008-4
- found SARS-CoV-2 RNA in the breath of infected individuals
- 2022 Leding et al. Detection of SARS-CoV-2 in exhaled breath from non-hospitalized COVID-19-infected individuals. https://www.nature.com/articles/s41598-022-15243-1
- found SARS-CoV-2 RNA in the breath of infected individuals
- We obtained 665 air samples from 111 included participants with confirmed SARS-CoV-2 infection. Overall, 52 individuals (46.8%) had at least one positive air sample, and 129 (19.4%) air samples were positive for SARS-CoV-2. Participants with symptoms or a symptom duration ≤ four days had significantly higher odds of having a positive air sample. Cycle threshold values were significantly lower in samples obtained ≤ 4 days from symptom onset. Neither variant of SARS-CoV-2 nor method of air sampling were associated with a positive air sample. We demonstrate that SARS-CoV-2 is detectable in human breath by electrostatic air sampling with the highest detection rate closest to symptom onset. We suggest further evaluation of the air sampling technique to increase sensitivity.
- Zhang et al. Monitoring SARS-CoV-2 in air and on surfaces and estimating infection risk …. April 27, 2022. https://www.nature.com/articles/s41370-022-00442-9
- SARS-CoV-2 was detected in the air.
- 2022-07. Parhizkar, Hooman, Leslie Dietz, Andreas Olsen-Martinez, Patrick F Horve, Liliana Barnatan, Dale Northcutt, and Kevin G Van Den Wymelenberg. ‘Quantifying Environmental Mitigation of Aerosol Viral Load in a Controlled Chamber With Participants Diagnosed With Coronavirus Disease 2019’. Clinical Infectious Diseases 75, no. 1 (1 July 2022): e174–84. https://doi.org/10.1093/cid/ciac006
- Had people live in a little modular room and measured the air.
- 2022-08. Moharir, Shivranjani C., Sharath Chandra Thota, Arushi Goel, Bhuwaneshwar Thakur, Dixit Tandel, S. Mahesh Reddy, Amareshwar Vodapalli, et al. “Detection of SARS-CoV-2 in the Air in Indian Hospitals and Houses of COVID-19 Patients.” Journal of Aerosol Science 164 (August 2022): 106002. https://doi.org/10.1016/j.jaerosci.2022.106002.
-
Taken together, we observed that the air around COVID-19 patients frequently showed the presence of SARS-CoV-2 RNA in both hospital and indoor residential settings and the positivity rate was higher when 2 or more COVID-19 patients occupied the room. In hospitals, SARS-CoV-2 RNA could be detected in ICUs as well as in non-ICUs, suggesting that the viral shedding happened irrespective of the severity of the infection. This study provides evidence for the viability of SARS-CoV-2 and its long-range transport through the air.
-
- 2022-08. Ramuta, Mitchell D., Christina M. Newman, Savannah F. Brakefield, et al. “SARS-CoV-2 and Other Respiratory Pathogens Are Detected in Continuous Air Samples from Congregate Settings.” Nature Communications 13, no. 1 (2022): 4717. https://doi.org/10.1038/s41467-022-32406-w.
- found SARS-CoV-2 RNA in school air.
- 2023-01. Gutmann et al. ‘Aerosol Measurement Identifies SARS-CoV 2 PCR Positive Adults …’. Env Res 216 (Jan 2023). https://www.sciencedirect.com/science/article/pii/S0013935122017443
- Measured SARS-CoV-2 particles out of COVID-positive and negative people. Many more out of +ve.
- Interesting:
-
“in SARS-CoV-2 PCR-positive group, 15.6% (n = 10/64) … were responsible for 64.8% of all exhaled particles counts in the group. Moreover, the 15.6%, equating to 3.5% of all patients (n = 10/288), was responsible for 51.2% of all exhaled particles.”
-
- 2021-07. Barbieri, Pierluigi, Luisa Zupin, Sabina Licen, et al. “Molecular Detection of SARS-CoV-2 from Indoor Air Samples in Environmental Monitoring Needs Adequate Temporal Coverage and Infectivity Assessment.” Environmental Research 198 (July 2021): 111200. https://doi.org/10.1016/j.envres.2021.111200.
- In the present work a short-term air monitoring was conducted at a geriatric COVID-19 healthcare facility in Trieste (Italy), where SARSCoV-2 RNA traces were detected. Evidence of viral RNA presence reported in Table 2 is coherent with those of the number of patients in the considered ward: the first day of the monitoring presents the highest number of patients and positive RNA finding in sampled indoor PM10.” (Barbieri et al., 2021, p. 4)
- Bazzazpour, Shahriyar, Masoumeh Rahmatinia, Seyed Reza Mohebbi, et al. “The Detection of SARS-CoV-2 RNA in Indoor Air of Dental Clinics during the COVID-19 Pandemic.” Environmental Science and Pollution Research International 29, no. 57 (2022): 85586–94. https://doi.org/10.1007/s11356-021-15607-6.
- Air sampling was done (n = 36) collecting particulate samples on PTFE filters at flow rates of 30 to 58 L/min. The samples were analyzed with novel coronavirus nucleic acid diagnostic real-time PCR kits. Only 13 out of 36 samples were positive for SARS-CoV-2 RNA. Logistic regression showed that sampling site’s volume, PM2.5 concentration, number of people, and number of active patient treatment units were significantly positively related with the presence of SARS-CoV-2 RNA. Thus, strategies to control the spread of COVID-19 should include reducing the number of infected people in dental clinics, adding filtration systems, and/or improving ventilation conditions.
- MacIntyre, C. Raina, Adriana Notaras, Noor Bari, et al. “Detection of SARS-CoV-2 in Aerosol and Surface Samples in High Acuity Hospital Settings during Community Epidemic Waves – Implications for Risk-Based Infection Control.” Respiratory Medicine 253 (March 2026). https://doi.org/10.1016/j.rmed.2026.108712.
- PCR only. Has a comment about fomite transmission being “prominent” which is quite surprising and I would look very closely at the 3 cited papers.
- I suspect the authors had to include this as a concession to the “droplet” enthusiasts on the author list.
other studies that found virus or its RNA in air samples, on air grates, and so on
- Coleman 2021. https://journals.plos.org/plospathogens/article?id=10.1371/journal.ppat.1009262
- Cheng 2021. https://academic.oup.com/cid/article/73/6/e1356/6225253
- more on air grills than high touch surfaces. See elsewhere.
- Chia 2020. https://www.nature.com/articles/s41467-020-16670-2
- Ge 2020. https://doi.org/10.1016/j.scs.2020.102413
- Jin 2020. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7428766/
- Kenarkoohi 2020. https://www.sciencedirect.com/science/article/abs/pii/S0048969720348531?via%3Dihub
- Lei 2020. https://onlinelibrary.wiley.com/doi/10.1111/irv.12783
- Liu 2020. https://www.nature.com/articles/s41586-020-2271-3
- Lednicky 2020. https://www.ijidonline.com/article/S1201-9712(20)30739-6/fulltext
- Ma 2020.
- Moore 2021. https://www.journalofhospitalinfection.com/article/S0195-6701(20)30548-X/fulltext
- a public health team conducted air sampling, generally negative but probably not done right. Still got 4/55 + samples
- Nissen 2020. https://www.nature.com/articles/s41598-020-76442-2
- RNA found in HEPA filters, 5 to 7 floors above COVID-positive patient areas, connected via ducts
- Razzini 2020. https://www.sciencedirect.com/science/article/pii/S0048969720340626?via%3Dihub
- Highest surface contamination on floor and air exhaust grates.
- Santarpia 2020. See above.
- Clarke, Rachel D., Nana Aisha Garba, Manuel A. Barbieri, et al. “Detection of SARS-CoV-2 in High-Efficiency Particulate Air (HEPA) Filters of Low-Cost Air Purifiers in Community-Based Organizations.” Environmental Monitoring and Assessment 195, no. 11 (2023): 1320. https://doi.org/10.1007/s10661-023-11950-y.
- Correia, Gil, Luís Rodrigues, Mariana Afonso, et al. “SARS-CoV-2 Air and Surface Contamination in Residential Settings.” Scientific Reports 12, no. 1 (2022): 18058. https://doi.org/10.1038/s41598-022-22679-y.
Coronavirus, SARS-1
- 2004-07. Agranovski, Igor E et al. ‘Monitoring of Viable Airborne SARS Virus in Ambient Air’. Atmospheric Environment 38, no. 23 (July 2004): 3879–84 https://www.sciencedirect.com/science/article/pii/S1352231004003292?via%3Dihub
- Isolated viable SARS-1 from the air
- Xiao. Detection of SARS-CoV and RNA on aerosol samples from SARS-patients admitted to hospital. https://pubmed.ncbi.nlm.nih.gov/15631748/
- detected SARS-1 genetic material in the air
- isolated live pathogen from one sample
-
Abstract
-
Objective: To assess the risk of aerosol transmission in severe acute respiratory syndrome (SARS) patients admitted to Hospital through testing the air samples.
-
Methods: Air samples were collected from 7 wards and 1 balcony of the Hospital, 3 times a day for 3 continuous days, using bioaerosol sampler type FA-2. Bioaerosol particles were then washed down from the samples by serum-free Dulbecco’s Modified Eagle Medium (DMEM) culture medium. Nested-reverse transcription-polymerase chain reaction (RT-PCR) was used to amplify the N protein gene of the SARS associated coronavirus (SARS-CoV) from these washing solutions. The residual solutions were inoculated into prepared cell cultures to isolate live virus. The positive samples were then identified by indirect immunofluorescence assay and sequence analysis of the PCR products.
-
Results: Positive rates of RT-PCR test on air samples were 29.03% in the wards and 20.0% in balcony respectively. Results from sequential analysis showed that the homology of amplified cDNA fragments to previously known SARS-CoV stains was 98%. A strain of live pathogen was isolated from one of the 36 samples. The isolate could cause typical cytopathic effects, similar to those SARS-CoV on Vero-E6 cells and the effects could be stably passed. Indirect immunofluorescence assay showed positive from serum of a SARS patient.
-
Conclusion: SARS-CoV existed in the air hospital, where SARS patients were admitted to, but the activity of SARS-CoV in air samples was rather low. SARS patients could still shed SARS-CoV even during the recovery phase. Potential possibility of aerosol transmission might exist within 1 meter square area around SARS patients.
- Booth, Timothy F et al. ‘Detection of Airborne Severe Acute Respiratory Syndrome (SARS) Coronavirus and Environmental Contamination in SARS Outbreak Units’. J. Infect. Dis. 191, no. 9 (1 May 2005): 1472–77. https://academic.oup.com/jid/article/191/9/1472/862003
- SARS found in air samples, confirmed by PCR, but authors were not able to culture replicating virus
- van Doremalen 2020. https://www.nejm.org/doi/full/10.1056/nejmc2004973
- Virus remained viable after 3 hours in the air without rapid decay despite high RH.
- SARS-1 as well as SARS-CoV-2
Coronavirus, MERS
- 2014-07. Azhar, Esam I et al. ‘Detection of the Middle East Respiratory Syndrome Coronavirus Genome in an Air Sample Originating from a Camel Barn Owned by an Infected Patient’. MBio, 22 July 2014. https://journals.asm.org/doi/10.1128/mbio.01450-14
- MERS, detected in air.
- Fellow had camels. Camels caught MERS. He caught MERS from camels.
- Authors found MERS RNA in an air sample from a barn.
- They could not culture viable virus.
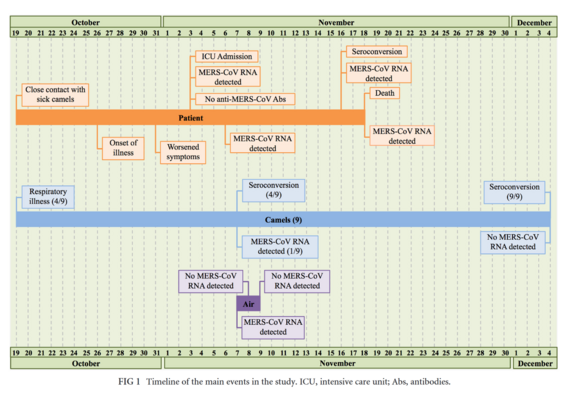
- 2014-12. Memish, Ziad A., Abdullah M. Assiri, and Jaffar A. Al-Tawfiq. ‘Middle East Respiratory Syndrome Coronavirus (MERS-CoV) Viral Shedding in the Respiratory Tract: An Observational Analysis with Infection Control Implications’. International Journal of Infectious Diseases 29 (December 2014): 307–8. https://doi.org/10.1016/j.ijid.2014.10.002
- MERS in respiratory secretions. Not in the air, but where do you think this implies it might also be, eh?
- 2016-02. Corman, Victor M., Ali M. Albarrak, Ali Senosi Omrani, Mohammed M. Albarrak, Mohamed Elamin Farah, Malak Almasri, Doreen Muth, et al. ‘Viral Shedding and Antibody Response in 37 Patients With Middle East Respiratory Syndrome Coronavirus Infection’. Clinical Infectious Diseases: An Official Publication of the Infectious Diseases Society of America 62, no. 4 (15 February 2016): 477–83. https://doi.org/10.1093/cid/civ951
- MERS in respiratory secretions
- “As in SARS, MERS-CoV nosocomial transmission was repeatedly ascribed to the potential of some patients to act as super-shedders or super-spreaders [6, 20]. Our analysis of viral loads, particularly in the early acute phase of disease, supports the existence of a limited number of patients with extraordinarily high viral loads. As these patients were not more likely to die of the infection, they might not have had more severe symptoms, and thus might have been able to engage in social contact despite their disease.”
- 2016-08. Kim, Sung-Han et al. ‘Extensive Viable Middle East Respiratory Syndrome (MERS) Coronavirus Contamination in Air and Surrounding Environment in MERS Isolation Wards’. Clin Inf Dise 63, no. 3 (1 August 2016): 363–69. https://academic.oup.com/cid/article/63/3/363/2595016
- Detour! Viable MERS virus in hospital air. 2016. Ya, they cultured it, you nay-saying ID doctor angling for a hospital promotion, you
Reports which suggest the spread of pathogen over more than two metres
Reports of outbreaks that occurred despite using contact precautions
other coronaviruses
- 1985 Mohammad Khalid Ijaz. Studies of the Airborne Survival of Rotaviruses and a Human Coronavirus. U of Ottawa, School of Med.
- Coronaviruses - suspended in air and viable 150 hours later. [posted July 2020]
- This thesis (1985) looked at effect of humidity & temp on aerosolized coronavirus. To keep the aerosol from depositing, put in drum rotating at 4 RPM. Not that fast.
- He cultured live virus up to 150 hours later. This is just an example of viral survival. These are Very artificial conditions. However, it demonstrates the virus can last some time in the air.
Filovirus (Ebola, etc.)
- .
Foot-and-mouth disease virus
- 2008 Gloster - foot-and-mouth … (talking about recovering viable aerosolized virus (viable) from pigs)
- 2008 Verreault - methods for sampling
- Christensen et al. “Detection of foot-and-mouth disease virus in the breath of infected cattle using a hand-held device to collect aerosols”. https://pubmed.ncbi.nlm.nih.gov/21723882/
- Animal studies are very useful because you can experimentally infect them. Here, they measured actual virus in cow breath.
- ““The estimated total release of airborne FMDV in 24 h from cattle is in the range of 10^4–10^5 TCID50 per 24 h depending on virus strains (Sellers and Parker, 1969; Donaldson et al., 1970; Alexandersen et al., 2002). Virus are shed in this way at a concentration of 1.0–1.5 log10 TCID50 per mL of exhaled air”
Hantavirus
-
Influenza
- 1962 Nature.
- Flu in the air. There are probably hundreds of articles over the 20th century about air spread. There’s not a single one for droplet by the way.

- 2009-02. Blachere, Francoise M, et al. ‘Measurement of Airborne Influenza Virus in a Hospital Emergency Department’. Clin. Infect. Dis. 48, no. 4 (15 February 2009): 438–40 https://academic.oup.com/cid/article/48/4/438/283890.
- Airborne influenza A detected in the air in emergency rooms (Blachere et al., 2009).
- 2016 . Leung, Nancy H. L., Jie Zhou, Daniel K. W. Chu, Han Yu, William G. Lindsley, Donald H. Beezhold, Hui-Ling Yen, et al. ‘Quantification of Influenza Virus RNA in Aerosols in Patient Rooms’. PloS One 11, no. 2 (2016): e0148669. https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0148669
- Airborne influenza A detected in the air in patient rooms (Leung et al., 2016).
- 2018-10. Traxler, Selina et al ‘VOC Breath Profile in Spontaneously Breathing Awake Swine during Influenza A Infection’. Scientific Reports 8, no. 1 (5 October 2018): 14857 https://www.nature.com/articles/s41598-018-33061-2.
- 2019-03. Bui, Vuong N et al. ‘Bioaerosol Sampling to Detect Avian Influenza Virus in Hanoi’s Largest Live Poultry Market’. Clinical Infectious Diseases 68, no. 6 (5 March 2019): 972–75. https://academic.oup.com/cid/article/68/6/972/5088836
- “The association between positive sample types (over time and position) was strong, with 91.7% of positive OP pooled swab samples confirmed by positive aerosol samples and 81% of influenza A positive aerosol samples confirmed by positive OP swab samples.”
- 2021-11. Dubuis, Marie-Eve et al. ‘High and Low Flowrate Sampling of Airborne Influenza in Hospital Rooms during Three Outbreaks’. Journal of Aerosol Science 158 (1 November 2021): 10582. https://www.sciencedirect.com/science/article/abs/pii/S0021850221005553?via%3Dihub
- La Rosa et al. Viral infections acquired indoors through airborne, droplet or contact transmission
- This article refers to multiple viruses sampled out of the air. There are some refs in this article about sampling flu out of the air and flu RNA, etc.
- Influenza virus found in breath in this:
-
… In clinical studies, virus-laden particles within the respirable aerosol fraction have been detected in exhaled breaths of patients with influenza and in the air samples from healthcare settings during seasonal peak [9]. Moreover, the scientific literature presents evidence in support of a contribution of aerosol transmission to the spread of influenza A, including the prolonged persistence of infectious aerosolized influenza virus at low humidity; the transmission of influenza by aerosols, reproducing the full spectrum of disease, at doses much smaller than those required by intranasal drop inoculation (large droplet transmission); and the interruption of transmission of in- fluenza by blocking the aerosol route through UV irradiation of upper room air [9-12]. A paper by Brankston and

-
- Found in air:
-
quences for hospitalized patients [14-16]. Using real-time polymerase chain reaction, Blachere and coworkers mea- sured the amount and size of airborne particles containing influenza virus in an emergency department. The authors confirmed the presence of airborne influenza virus, and found over 50% of detectable influenza virus particles to be within the respirable aerosol fraction [10]. Lindsley and

-
- Found flu RNA in air:
-
be within the respirable aerosol fraction [10]. Lindsley and colleagues detected small airborne particles containing in- fluenza RNA in a health care facility during influenza sea- son. They also found a correlation between the number of influenza-positive samples and the number and location of patients with influenza [17]. As for contact transmis-

-
- Probably aint yer hands, you dirty monster bros:
-
influenza season [ 18, 19]. Viruses can be transferred from surfaces to hands, and vice versa. The importance of this mode of transmission for influenza is unclear however, since, while the virus can survive on surfaces for hours or even days, it cannot survive on hands for longer than five minutes [20]. A recent study concluded that influenza A transmission via fomites is possible but unlikely to occur [19]. The overall burden of health care facility-acquired in-

-
- Thread about the La Rose article (from 2013) is here, listing many viruses found in the air over the years, including coronaviruses, influenza, adenovirus, etc. https://mastodon.social/@jmcrookston/111994168869167484
studies where viable pathogen was isolated out of the air in a laboratory setting
- 2009 . Fabian et al (Milton) - An optimized method to … (detected viable flu from air)
- https://pubmed.ncbi.nlm.nih.gov/19689447/
- viable pathogen was isolated out of the air in a laboratory setting
- Milton 2013. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3591312/
- viable pathogen was isolated out of the air in a laboratory setting
- 2015 . Lindsley, William G. et al. ‘Viable Influenza A Virus in Airborne Particles from Human Coughs’. Journal of Occupational and Environmental Hygiene 12, no. 2 (2015): 107–13. https://www.tandfonline.com/doi/full/10.1080/15459624.2014.973113 or https://pubmed.ncbi.nlm.nih.gov/25523206/
- Influenza and viable virus again.
- 2016-09. Lindsley, William G. et al. ‘Viable Influenza A Virus in Airborne Particles Expelled during Coughs versus Exhalations’. Influenza and Other Respiratory Viruses 10, no. 5 (September 2016): 404–13. https://onlinelibrary.wiley.com/doi/10.1111/irv.12390
- Influenza and viable virus.
- Pan 2017. https://journals.asm.org/doi/10.1128/mSphere.00251-17
- 2018-01. Yan, Jing ‘Infectious Virus in Exhaled Breath of Symptomatic Seasonal Influenza Cases from a College Community’. Proceedings of the National Academy of Sciences 115, no. 5 (30 January 2018): 1081–86. https://www.pnas.org/doi/full/10.1073/pnas.1716561115 or https://www.pnas.org/content/115/5/1081
- Influenza. Found viable virus in air.

- Leung 2020. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8238571/
- viable pathogen was isolated out of the air in a laboratory setting
Measles
virus remained viable in air over a significant period of time
- De Jong 1964 https://www.nature.com/articles/2011054a0
- found viable virus in the air after 2 hours. Less decay at low RH versus high RH.
- Sattar, Syed A., Mohammad Khalid Ijaz, and Charles P. Gerba. “Spread of Viral Infections by Aerosols.” Critical Reviews in Environmental Control 17, no. 2 (1987): 89–131. https://doi.org/10.1080/10643388709388331.
- See above.
- Bischoff 2016 https://academic.oup.com/jid/article/213/4/600/2459431
- found genetic material of the virus in aerosol samples or in places which would not be touched
Norovirus
- Bonifait et al. ‘Detection and Quantification of Airborne Norovirus during Outbreaks in Healthcare Facilities’. Clin Inf Dis: 61, no. 3 (1 August 2015): 299–304. https://academic.oup.com/cid/article/61/3/299/491373
- Cheng, V. C. C., S.-C. Wong, K. H. Y. Chiu, C. C. Y. Yip, S. C. Y. Wong, and K.-Y. Yuen. “Detection of Norovirus in Air Samples in a Non-Vomiting Patient: Implications of Testing Saliva for Norovirus in an Immunocompromised Host.” The Journal of Hospital Infection 103, no. 3 (November 2019): 357–58. https://doi.org/10.1016/j.jhin.2019.07.011.
- Marks, P. J., I. B. Vipond, F. M. Regan, K. Wedgwood, R. E. Fey, and E. O. Caul. “A School Outbreak of Norwalk-like Virus: Evidence for Airborne Transmission.” Epidemiology and Infection 131, no. 1 (August 2003): 727–36.
-
“As each area where a child vomited was cleaned immediately after vomiting had occurred, spread must have been by airborne transmission directly to susceptible individuals, or via contamination of the wider environment.” Marks et al., 2003, p. 733
-
“It also produced further evidence to suggest that aerosolization of virus particles can lead to direct infection.” Marks et al., 2003, p. 735
-
“this study supports laboratory evidence that cleaning with quaternary ammonium products is unlikely to alter the course of an outbreak.” Marks et al., 2003, p. 735
-
Francisella tularensis (bacteria)
- Quantification of the Relationship between Bacterial Kinetics and Host Response for Monkeys Exposed to Aerosolized Francisella tularensis. https://journals.asm.org/doi/full/10.1128/aem.01190-10
- “Francisella tularensis can be disseminated via aerosols, and once inhaled, only a few microorganisms may result in tularemia pneumonia.”
- (This is really a quantification paper. I need a quantification thread.)
- Written by Huang and Haas
Tuberculosis (bacteria)
viable pathogen recovered from the air
- 2003-07. Menzies, Dick, Neill Adhikari, Marie Arietta, and Vivian Loo. ‘Efficacy of Environmental Measures in Reducing Potentially Infectious Bioaerosols During Sputum Induction’. Infection Control & Hospital Epidemiology 24, no. 7 (July 2003): 483–89. https://doi.org/10.1086/502242
- TB in air rapidly climbed when you induced sputum. Overwhelmed ventilation.
- Detected TB by plates. This means the bacteria was cultured for all the fantastic, absolutely miserable, skeptic ID/IPAC practitioners out there.
- Fennelly 2003. https://pubmed.ncbi.nlm.nih.gov/14656754/
- Fennelly 2012. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3443801/
- Patterson 2018. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5757796/
- Theron 2020. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8353872/
- Dinkele 2021. https://journals.plos.org/plospathogens/article?id=10.1371/journal.ppat.1009262
unsorted
- 1962-04. Riley, R L, C C Mills, F O’Grady, L U Sultan, F Wittstadt, and D N Shivpuri. ‘Infectiousness of Air from a Tuberculosis Ward. Ultraviolet Irradiation of Infected Air: Comparative Infectiousness of Different Patients’. The American Review of Respiratory Disease 85 (April 1962): 511–25. https://doi.org/10.1164/arrd.1962.85.4.511
- The famous study that showed TB was in air. Patients’ rooms connected to guinea pigs by vents. Guinea pigs would get infected over time.
- 1988-05. Kantor et al. ‘Nosocomial Transmission of Tuberculosis from Unsuspected Disease’. The American Journal of Medicine 84, no. 5 (May 1988): 833–38. https://doi.org/10.1016/0002-9343(88)90060-5
- Not about detecting it in air, but here 9 of 56 people working in a hospital caught TB from a patient who hadn’t been diagnosed with it. He seemed to infect 5 during his time on the ward before he died.
- His bacteria riddled lungs infected the next 4, in the autopsy room, when they were compressed during the post-mortem
- 2007-05. Escombe, A. Roderick, Clarissa Oeser, Robert H. Gilman, Marcos Navincopa, Eduardo Ticona, Carlos Martínez, Luz Caviedes, et al. ‘The Detection of Airborne Transmission of Tuberculosis from HIV-Infected Patients, Using an in Vivo Air Sampling Model’. Clinical Infectious Diseases: An Official Publication of the Infectious Diseases Society of America 44, no. 10 (15 May 2007): 1349–57. https://doi.org/10.1086/515397
- Transmission TB through the air from people with HIV. HIV infects the lungs too.
- 2012-05. Dharmadhikari et al. ‘Surgical Face Masks Worn by Patients with Multidrug-Resistant Tuberculosis: Impact on Infectivity of Air on a Hospital Ward’. American Journal of Respiratory and Critical Care Medicine 185, no. 10 (15 May 2012): 1104–9. https://doi.org/10.1164/rccm.201107-1190OC
- Patients wearing surgical masks lowered spread
-
“Methods: Over 3 months, 17 patients with pulmonary MDR-TB occupied an MDR-TB ward in South Africa and wore face masks on alternate days. Ward air was exhausted to two identical chambers, each housing 90 pathogen-free guinea pigs that breathed ward air either when patients wore surgical face masks (intervention group) or when patients did not wear masks (control group). Efficacy was based on differences in guinea pig infections in each chamber….”
-
“… Results: Sixty-nine of 90 control guinea pigs (76.6%; 95% confidence interval [CI], 68–85%) became infected, compared with 36 of 90 intervention guinea pigs (40%; 95% CI, 31–51%), representing a 56% (95% CI, 33–70.5%) decreased risk of TB transmission when patients used masks. Conclusions: Surgical face masks on patients with MDR-TB significantly reduced transmission and offer an adjunct measure for reducing TB transmission from infectious patients.”
- 2022-07. Dinkele et al ‘Aerosolization of Mycobacterium Tuberculosis by Tidal Breathing’. American Journal of Respiratory and Critical Care Medicine 206, no. 2 (15 July 2022): 206–16. https://doi.org/10.1164/rccm.202110-2378OC
- Tidal breathing spreads TB bacteria. And while coughs produce more TB per cough, their infrequency means tidal breathing contributes more to exhalation than cough.
strep (bacteria)
- 1938 . Strep in air. [originally posted Jan 13, 2021]
-
“… the fact that 22 patients..actively inspiring strep-laden air @ 13.7 cubic ft/hour… & adding to air infection by coughing & sneezing … it does not need much stretch of imagination to accept view that infection by particles in the air is no chimera.”

- Strep is a bacteria - bigger than virus. Covs are ~0.120uM. Strep is 0.5 to 2.0uM say. More than ten times bigger. Should be even more prone to “fall quickly to the ground” to people who said that.

-
other pathogens
- 2010 . Bacteria in air of turkey plant. https://pubmed.ncbi.nlm.nih.gov/20720091/
- 2011 . More pathogens in the air of a turkey plant. https://pubmed.ncbi.nlm.nih.gov/21170464/
- Authors cultured some.
- 2011 . Review article about airborne pathogens emitted from sewage treatment plants. https://pubmed.ncbi.nlm.nih.gov/21196384/
- Alonso et al. Evidence of infectivity of airborne porcine epidemic diarrhea virus and detection of airborne virua RNA at long distances from infected herds

Studies showing that a pathogen can infect over a distance (i.e. where animals infected each other by airborne spread)
- 1941 ferret study showed that influenza transmitted from ferret to ferret 5 feet over, and up, so it could not be droplets.
- 2002-11. Brockmeier, Susan L, and Kelly M Lager. “Experimental Airborne Transmission of Porcine Reproductive and Respiratory Syndrome Virus and Bordetella Bronchiseptica.” Veterinary Microbiology 89, no. 4 (November 6, 2002): 267–75. https://doi.org/10.1016/S0378-1135(02)00204-3.
-
“Experiments were designed to determine if porcine reproductive and respiratory syndrome virus (PRRSV) or Bordetella bronchiseptica could be transmitted through indirect airborne contact” ([Brockmeier and Lager, 2002, p. 267]
-
…
-
“Airborne transmission of B. bronchiseptica occurred in 5/5 trials where B. bronchiseptica was the only agent used, and in 3/5 trials where the principal pigs were coinfected with both agents. Airborne transmission of PRRSV occurred in 4/5 trials where PRRSV was the only agent used, and in 2/5 trials where the principal pigs were coinfected with both agents. Thus, airborne transmission of both agents over short distances, such as within a barn, is probable.” ([Brockmeier and Lager, 2002, p. 267]
-
- 2010-10. McCullers, Jonathan A., Julie L. McAuley, Sarah Browall, Amy R. Iverson, Kelli L. Boyd, and Birgitta Henriques Normark. “Influenza Enhances Susceptibility to Natural Acquisition of and Disease Due to Streptococcus Pneumoniae in Ferrets.” The Journal of Infectious Diseases 202, no. 8 (October 15, 2010): 1287–95. https://doi.org/10.1086/656333.
-
“We also did not assess the mode of transmission. It is likely that aerosol transmission was required for acquisition by contact ferrets placed 3.5 m away, because large droplets are unlikely to be capable of crossing this space, particularly in a room with such a high rate of air exchange. Sneezing by ferrets infected with influenza virus may have contributed either to formation of aerosols or dissemination across that distance. However, contacts infected with influenza virus were capable of acquiring pneumococcus from donors 3.5 m away that were not infected with influenza and were not sneezing.” ([McCullers et al., 2010, p. 1293]
-
Animal model studies. These either infect animals by air, isolate pathogen out of animal breath, or show that animals can infect each other through the air
Coronaviruses, SARS
- Kutter 2021. https://www.nature.com/articles/s41467-021-21918-6
- SARS
Coronaviruses, MERS
- Totura 2020. https://pubmed.ncbi.nlm.nih.gov/32744989/
- MERS
Coronaviruses, SARS-CoV-2
- Richard 2020. https://www.nature.com/articles/s41467-020-17367-2
- ferrets
- Sia 2020. https://www.nature.com/articles/s41586-020-2342-5
- hamsters
- Kutter 2021. https://www.nature.com/articles/s41467-021-21918-6
- ferrets
- Port 2021. https://www.nature.com/articles/s41467-021-25156-8
- hamsters
- Yinda 2021. https://pubmed.ncbi.nlm.nih.gov/34120579/
- hamsters
- Dabisch 2021. https://journals.plos.org/plospathogens/article?id=10.1371/journal.ppat.1009865
- monkeys
- Zhang 2021. https://wwwnc.cdc.gov/eid/article/27/7/20-3948_article
- monkeys
Filoviruses
situations of animals spreading, or strongly suggesting animal spread by air
- Jaax, N., P. Jahrling, T. Geisbert, et al. “Transmission of Ebola Virus (Zaire Strain) to Uninfected Control Monkeys in a Biocontainment Laboratory.” Lancet (London, England) 346, nos. 8991–8992 (1995): 1669–71. https://doi.org/10.1016/s0140-6736(95)92841-3.
- Ebolavirus spread to monkeys over 2 metres
- Weingartl, Hana M., Carissa Embury-Hyatt, Charles Nfon, Anders Leung, Greg Smith, and Gary Kobinger. “Transmission of Ebola Virus from Pigs to Non-Human Primates.” Scientific Reports 2, no. 1 (2012): 811. https://doi.org/10.1038/srep00811.
- Wong, Gary, Xiangguo Qiu, Jason S. Richardson, et al. “Ebola Virus Transmission in Guinea Pigs.” Journal of Virology 89, no. 2 (2014): 1314–23. https://doi.org/10.1128/jvi.02836-14.
experimentally infecting animals via aerosols
- Alves, D. A., A. R. Glynn, K. E. Steele, et al. “Aerosol Exposure to the Angola Strain of Marburg Virus Causes Lethal Viral Hemorrhagic Fever in Cynomolgus Macaques.” Veterinary Pathology 47, no. 5 (2010): 831–51. https://doi.org/10.1177/0300985810378597.
- Johnson, E., N. Jaax, J. White, and P. Jahrling. “Lethal Experimental Infections of Rhesus Monkeys by Aerosolized Ebola Virus.” International Journal of Experimental Pathology 76, no. 4 (1995): 227–36.
influenza
- Andrewes 1941. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2065394/
- ferrets
- Schulman 1962. https://www.nature.com/articles/1951129a0
- mice
- Lowen 2006. https://pubmed.ncbi.nlm.nih.gov/16785447/
- guinea pigs
- Mubareka 2009. https://pubmed.ncbi.nlm.nih.gov/19434931/
- guinea pigs
- Koster 2012. https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0033118
- ferrets
tuberculosis
- GUINEA PIGS:
- Alsaadi 1973. https://pubmed.ncbi.nlm.nih.gov/4197232/
- Riley 1959. https://academic.oup.com/aje/article-abstract/70/2/185/182026
- Riley 1961. https://www.atsjournals.org/doi/abs/10.1164/arrd.1962.85.4.511
- Chambers 2002. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC127856/
- Escombe 2007. https://academic.oup.com/cid/article/44/10/1349/356240
- Escombe 2009. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2656548/
- Dharmadhikari 2012. https://pubmed.ncbi.nlm.nih.gov/22323300/
Review articles and reports of airborne spread between or in buildings
review articles
- Shen, Jialei, Meng Kong, Bing Dong, Michael J. Birnkrant, and Jianshun Zhang. “Airborne Transmission of SARS-CoV-2 in Indoor Environments: A Comprehensive Review.” Science and Technology for the Built Environment 27, no. 10 (2021): 1331–67. https://doi.org/10.1080/23744731.2021.1977693.
reports of spread between buildings and between units in buildings
- Gao, Naiping, Jianlei Niu, Marco Perino, and Per Heiselberg. ‘The Airborne Transmission of Infection between Flats in High-Rise Residential Buildings: Particle Simulation’. Building and Environment 44 (1 February 2009): 402–10. https://doi.org/10.1016/j.buildenv.2008.03.016.
- Gao, N.P., J.L. Niu, M. Perino, and P. Heiselberg. ‘The Airborne Transmission of Infection between Flats in High-Rise Residential Buildings: Tracer Gas Simulation’. Building and Environment 43, no. 11 (November 2008): 1805–17. https://doi.org/10.1016/j.buildenv.2007.10.023.
- Huang, Jianxiang, Phil Jones, Anqi Zhang, Shan Shan Hou, Jian Hang, and John D. Spengler. ‘Outdoor Airborne Transmission of Coronavirus Among Apartments in High-Density Cities’. Frontiers in Built Environment 7 (2021). https://doi.org/10.3389/fbuil.2021.666923.
- Wei, Hsin-Yi, Cheng-Ping Chang, Ming-Tsan Liu, Jung-Jung Mu, Yu-Ju Lin, Yu-Tung Dai, and Chia-Ping Su. ‘Probable Aerosol Transmission of SARS-CoV-2 through Floors and Walls of Quarantine Hotel, Taiwan, 2021’. Emerging Infectious Diseases 28, no. 12 (December 2022): 2374–82. https://doi.org/10.3201/eid2812.220666.
- Zheng, Jianwen, Qiuhua Tao, and Yangui Chen. ‘Airborne Infection Risk of Inter-Unit Dispersion through Semi-Shaded Openings: A Case Study of a Multi-Storey Building with External Louvers’. Building and Environment 225 (1 November 2022): 109586. https://doi.org/10.1016/j.buildenv.2022.109586.
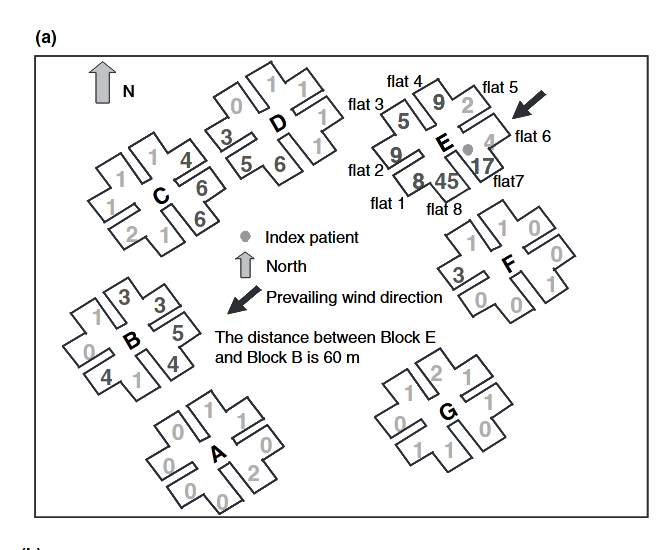
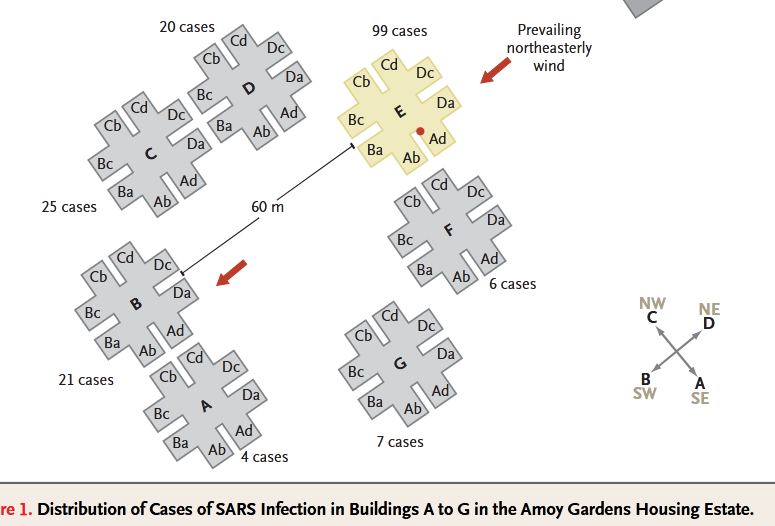
- Li, Y., S. Duan, I. T. S. Yu, and T. W. Wong. “Multi-Zone Modeling of Probable SARS Virus Transmission by Airflow between Flats in Block E, Amoy Gardens.” Indoor Air 15, no. 2 (April 2005): 96–111. https://doi.org/10.1111/j.1600-0668.2004.00318.x.
- Once more for the people at the back, SARS TRANSMITTED BETWEEN APARTMENT BLOCKS AND DOWN THE STREET.
- Diagram of six apartment blocks, showing where people caught SARS-1 and that they were downwind of the index patient.
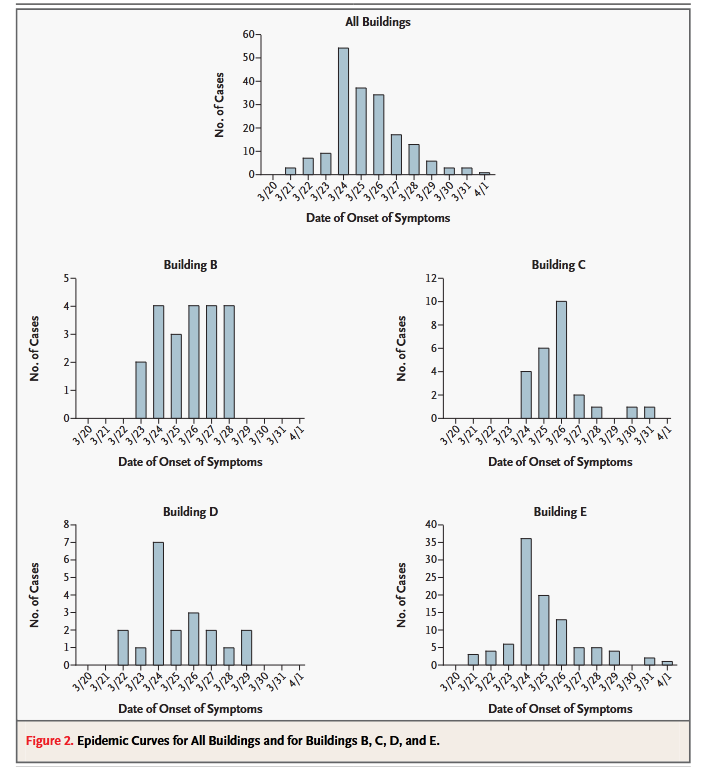
- Yu, Ignatius T S, Yuguo Li, Tze Wai Wong, Wilson Tam, Andy T Chan, Joseph H W Lee, Dennis Y C Leung, and Tommy Ho. “Evidence of Airborne Transmission of the Severe Acute Respiratory Syndrome Virus.” N. Engl. J. Med. 350, no. 17 (April 22, 2004): 1731–39. https://doi.org/10.1056/NEJMoa032867.
- Diagram of 7 buildings in apartment complex and prevailing wind direction. Infections occurred in downwind buildings.
- Onset charts of when people got sick in four buildings at Amoy Gardens.
- Diagram of 7 buildings in apartment complex and prevailing wind direction. Infections occurred in downwind buildings.
When Chapin in his 1910 book The Sources and Modes of Infection said “airborne”, he meant over a couple hundred metres to a couple of kilometres. Oh, by the way, in Amoy Gardens, an apartment block, SARS spread over … a couple hundred metres, in the direction of the prevailing wind.
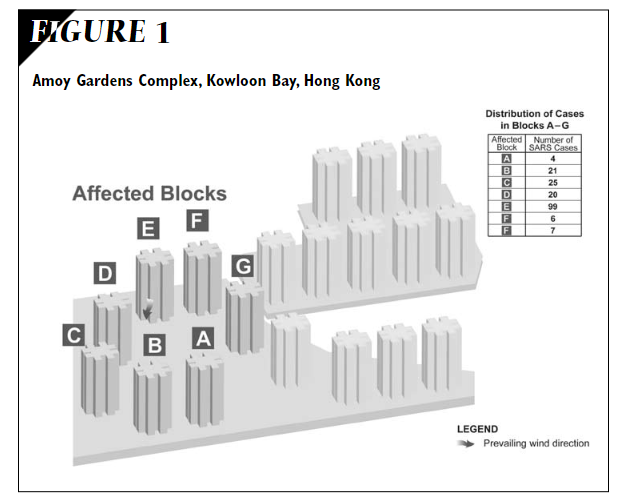
- “The epidemiological data curve for the Amoy Gardens cases was consistent with that for an outbreak with a common source (Yu et al., 2004)”
- https://neha.org/sites/default/files/jeh/JEH5.06-Feature-Environmental-Transmission-of-SARS.pdf
reports of spread through building systems
- Gormley, Michael, Thomas J. Aspray, David A. Kelly, and Cristina Rodriguez-Gil. ‘Pathogen Cross-Transmission via Building Sanitary Plumbing Systems in a Full Scale Pilot Test-Rig’. PLoS ONE 12, no. 2 (10 February 2017). https:// doi.org/10.1371/journal.pone.0171556.
- Kang, Min, Jianjian Wei, Jun Yuan, Juxuan Guo, Yingtao Zhang, Jian Hang, Yabin Qu, et al. ‘Probable Evidence of Fecal Aerosol Transmission of SARS-CoV-2 in a High-Rise Building’. Annals of Internal Medicine 173, no. 12 (15 December 2020): 974–80. https://doi.org/10.7326/M20-0928.
- Gormley, Michael, Thomas J. Aspray, and David A. Kelly. ‘Aerosol and Bioaerosol Particle Size and Dynamics from Defective Sanitary Plumbing Systems’. Indoor Air 31, no. 5 (2021): 1427–40. https://
doi.org/10.1111/ina.12797.
- Modelling transmission through defective sanitary plumbing systems.
- Du, Chun-Ru, Shun-Chih Wang, Ming-Chih Yu, Ting-Fang Chiu, Jann-Yuan Wang, Pei-Chun Chuang, Ruwen Jou, Pei-Chun Chan, and Chi-Tai Fang. ‘Effect of Ventilation Improvement during a Tuberculosis Outbreak in Underventilated University Buildings’. Indoor Air 30, no. 3 (May 2020): 422–32. https://doi.org/10.1111/ina.12639.
- I’ll just highlight this paper, decreasing CO2 from 3000 to <1000 cut TB cases by 97%
-
“After adjusting for effects of contact investigation and latent TB infection treatment, improving ventilation rate to levels with CO2 <1000 ppm was independently associated with a 97% decrease (95% CI:50%-99.9%) in the incidence of TB among contacts. “
- Nissen, Karolina, Janina Krambrich, Dario Akaberi, Tove Hoffman, Jiaxin Ling, Åke Lundkvist, Lennart Svensson, and Erik Salaneck. “Long-Distance Airborne Dispersal of SARS-CoV-2 in COVID-19 Wards.” Scientific Reports 10, no. 1 (November 11, 2020): 19589. https:// doi.org/10.1038/s41598-020-76442-2.
- Ding, Shirun, Zhen Wei Teo, Man Pun Wan, and Bing Feng Ng. “Aerosols from Speaking Can Linger in the Air for up to Nine Hours.” Building and Environment 205 (November 2021): 108239. https://
doi.org/10.1016/j.buildenv.2021.108239.
- The aerosols were found to linger in air up to 9 hours (not strictly speaking on topic for this thread, but highly relevant)
- Doremalen, Neeltje van, Trenton Bushmaker, Dylan H. Morris, Myndi G. Holbrook, Amandine Gamble, Brandi N. Williamson, Azaibi Tamin, et al. “Aerosol and Surface Stability of SARS-CoV-2 as Compared with SARS-CoV-1.” New England Journal of Medicine 382, no. 16 (April 16, 2020): 1564–67. https://doi.org/10.1056/NEJMc2004973.
-
“SARS-CoV-2 remained viable in aerosols throughout the duration of our experiment (3 hours), with a reduction in infectious titer from 103.5 to 102.7 TCID50 per liter of air. This reduction was similar to that observed with SARS-CoV-1, from 104.3 to 103.5 TCID50 per milliliter (Fig. 1A).”
-
- Fears, Alyssa C., William B. Klimstra, Paul Duprex, Amy Hartman, Scott C. Weaver, Kenneth S. Plante, Divya Mirchandani, et al. “Persistence of Severe Acute Respiratory Syndrome Coronavirus 2 in Aerosol Suspensions - Volume 26, Number 9—September 2020 - Emerging Infectious Diseases Journal - CDC,” September 2020. https://doi.org/10.3201/eid2609.201806.
-
“We aerosolized severe acute respiratory syndrome coronavirus 2 and determined that its dynamic aerosol efficiency surpassed those of severe acute respiratory syndrome and Middle East respiratory syndrome coronaviruses. Although we performed experiments only once in each of several laboratories, our findings suggest retained infectivity and virion integrity for up to 16 hours in respirable-sized aerosols.” (Fears et al., 2020, p. 2168)”
-
When Chapin in his 1910 book The Sources and Modes of Infection said “airborne”, he meant over a couple hundred metres to a couple of kilometres.
Oh, by the way, in Amoy Gardens, an apartment block, SARS spread over … a couple hundred metres, in the direction of the prevailing wind.
Pathogen cross-transmission via building sanitary plumbing systems in a full scale pilot test-rig The WHO Consensus Document on the epidemiology of the SARS epidemic in 2003, included a report on a concentrated outbreak in one Hong Kong housing block which was considered a ‘super-spreading event’. The WHO report conjectured that the sanitary plumbing system was one transmission route for the virus. Empty U-traps allowed the aerosolised virus to enter households from the sewerage system. No biological evidence was presented. This research reports evidence that pathogens can be aerosolised and transported on airstreams within sanitary plumbing systems and enter buildings via empty U-traps. A sanitary plumbing system was built, representing two floors of a building, with simulated toilet flushes on the lower floor and a sterile chamber with extractor fan on the floor above. Cultures of a model organism, Pseudomonas putida at 106–109 cfu ml-1 in 0·85% NaCl were flushed into the system in volumes of 6 to 20 litres to represent single or multiple toilet flushes. Air and surface samples were cultured on agar plates and assessed qualitatively and semi-quantitatively. Flushing from a toilet into a sanitary plumbing system generated enough turbulence to aerosolise pathogens. Typical sanitary plumbing system airflows (between 20–30 ls-1) were sufficient to carry aerosolised pathogens between different floors of a building. Empty U-traps allowed aerosolised pathogens to enter the chamber, encouraging cross-transmission. All parts of the system were found to be contaminated post-flush. Empty U-traps have been observed in many buildings and a risk assessment indicates the potential for high risk cross-transmission under defect conditions in buildings with high pathogen loading such as hospitals. Under defective conditions (which are not uncommon) aerosolised pathogens can be carried on the airflows within sanitary plumbing systems. Our findings show that greater consideration should be given to this mode of pathogen transmission. May 9, 2024 Jonathan @jmcrookston
All other reports of the airborne transmission of pathogens, sorted by pathogen
In the pinned posts here, https://mastodon.social/@jmcrookston/110861950615588941, you will find threads about various pathogens in the air, including:
- TB
- measles
- chickenpox
- animal coronaviruses
- rhinovirus
- filoviruses
- MRSA
- lassavirus
- leprosy
I will start moving these here.
chickenpox
Reports which suggest the spread of pathogen over more than two metres
- Leclair 1980 - see above
- Asano 1980. https://pubmed.ncbi.nlm.nih.gov/6257228/
- Josephson 1988. https://pubmed.ncbi.nlm.nih.gov/3392417/
- Gustafson 1982. https://pubmed.ncbi.nlm.nih.gov/6289235/
Outbreak investigations with infection despite using contact and droplet precautions
- Leclair 1980 - see above
- Asano 1980 - see above
- Anderson 1985. https://pubmed.ncbi.nlm.nih.gov/3845064/
- Josephson 1988 - see above
- Menkhaus 1990. https://www.thelancet.com/journals/lancet/article/PII0140-6736(90)92996-U/fulltext
- Gustafson 1982 - see above
coronaviruses, animal coronaviruses
- Animal coronaviruses are known to transmit by air. https://mastodon.social/@jmcrookston/111467649735806761
coronaviruses, SARS and MERS
virus remained viable in air over a significant period of time
- van Doremalen 2020. See above.
Reports which suggest the spread of pathogen over more than two metres
- Christian, Michael D., Mona Loutfy, L. Clifford McDonald, et al. ‘Possible SARS Coronavirus Transmission during Cardiopulmonary Resuscitation’. Emerging Infectious Diseases 10, no. 2 (February 2004): 287–93. https://wwwnc.cdc.gov/eid/article/10/2/03-0700_article. https://doi.org/10.3201/eid1002.030700.
- It was in the room air, not because of an aerosol generating medical procedure, of course, but they wanted to reduce “panic” so they lied and said it was droplet+AGMP.
- 2004-04. Yu, Ignatius T S, Yuguo Li, Tze Wai Wong, Wilson Tam, Andy T Chan, Joseph H W Lee, Dennis Y C Leung, and Tommy Ho. ‘Evidence of Airborne Transmission of the Severe Acute Respiratory Syndrome Virus’. N. Engl. J. Med. 350, no. 17 (22 April 2004): 1731–39. https://www.nejm.org/doi/full/10.1056/NEJMoa032867
- Olsen 2003. https://pubmed.ncbi.nlm.nih.gov/14681507/
- Yu 2004. https://pubmed.ncbi.nlm.nih.gov/15102999/
- Li 2005. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3591312/
- Li, Y., X. Huang, I. T. S. Yu, T. W. Wong, and H. Qian. ‘Role of Air Distribution in SARS Transmission during the Largest Nosocomial Outbreak in Hong Kong’. Indoor Air 15, no. 2 (April 2005): 83–95. https://onlinelibrary.wiley.com/doi/10.1111/j.1600-0668.2004.00317.x
- air modelling matched the infecteds
- Amoy Gardens
- And everything written about Amoy Gardens, showing the spread between buildings, caused by negative pressure sucking air up the sewage stacks and then (a) into units (b) in plumes to buildings downwind…
Reports of outbreaks that occurred despite using contact precautions
- Christian, see above.
- Li 2004. https://onlinelibrary.wiley.com/doi/10.1111/j.1600-0668.2004.00317.x
- Yu 2004. https://pubmed.ncbi.nlm.nih.gov/15102999/
- Wong 2004. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3322939/
coronaviruses, SARS-COV-2/COVID-19
virus remained viable in air over a significant period of time
- van Doremalen 2020. See above.
Reports which suggest the spread of pathogen over more than two metres
- Azimi 2021. https://www.pnas.org/content/118/8/e2015482118
- Eichler 2021. https://wwwnc.cdc.gov/eid/article/27/5/21-0514_article
- Cai 2020. https://pubmed.ncbi.nlm.nih.gov/32163030/
- Günther 2020. https://www.embopress.org/doi/full/10.15252/emmm.202013296
- Miller 2020. https://onlinelibrary.wiley.com/doi/10.1111/ina.12751
- Jang 2020. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7392463/
- Li 2021. https://www.sciencedirect.com/science/article/abs/pii/S0360132321001955
- Katelaris 2021. https://wwwnc.cdc.gov/eid/article/27/6/21-0465_article
- Cheng 2021. See elsewhere.
- too many more to mention, but I will mention them. Eventually.
Reports of outbreaks that occurred despite using “contact and droplet” precautions
- Klompas 2021. https://www.acpjournals.org/doi/full/10.7326/M20-7567
- Klompas 2021. https://academic.oup.com/cid/advance-article/doi/10.1093/cid/ciab218/6168040
- Goldberg 2021. https://academic.oup.com/ofid/article/8/3/ofab036/6121257?login=true
- Oksanen 2021. https://pubmed.ncbi.nlm.nih.gov/33847307/
- Cheng 2021. See elsewhere.
- Karan 2021. https://academic.oup.com/cid/advance-article/doi/10.1093/cid/ciab564/6305137
- Saidel-Odes 2021. https://pubmed.ncbi.nlm.nih.gov/33432896/
human challenge trials
- Human challenge, the subjects infected by intranasal drops. However, measured who spread (2 of 34 responsible for most spread), how long (3 days), and measured virus in air and in breath.
Filoviruses (Marburgvirus, Ebolavirus, Restonvirus, etc.)
- thread https://mastodon.social/@jmcrookston/111145873500577577
-
review articles discussing possibility of airborne spread
- 2015-01. Ponce De Leon-Rosales, et al. ‘Ebola, Through Air or Not Through Air: That Is the Question’. Frontiers in Public Health 2 (19 January 2015): 292. https://doi.org/10.3389/fpubh.2014.00292
- Possible that the dose is very low and transmission by air might be possible even if droplet precautions are used.
- “If we consider that the infectious dose may be very low – at only 10 virus particles (3) – and that an airborne particle may contain thousands of Ebola virions, thus an exposure carried by air can be risky.”
- 2016-05. Mekibib, Berhanu, and Kevin K Ariën. ‘Aerosol Transmission of Filoviruses’. Viruses 8, no. 5 (23 May 2016). https://doi.org/10.3390/v8050148
- This review article dealt with that question. Authors noted:
- Ebolavirus shed from mouth and nasal cavities of pigs.
- Ebola in bronchial washings of monkeys after aerosol inoculation
- Copious extracellular EBOV antigen on mucosal surfaces of the nose when exposed to infected air.
- “At the very least, the potential exists for aerosol transmission, given that virus is detected in bodily secretions, the pulmonary alveolar interstitial cells, and within lung spaces.”
- This review article dealt with that question. Authors noted:
Hantaviruses
- 2008-03. Pettersson, Lisa ‘Hantavirus RNA in Saliva from Patients with Hemorrhagic Fever with Renal Syndrome’. Emerging Infectious Diseases 14, no. 3 (March 2008): 406–11 https://wwwnc.cdc.gov/eid/article/14/3/07-1242_article
- Which other do you want me to do? For fun, hantavirus in saliva. You know what that probably means …
- Pinna. 2004. “New epidemiologic and molecular evidence of person to person transmission of hantavirus Andes Sout…”
-
“After the Hantavirus Pulmonary Syndrome (HPS) outbreak occurred in Southern Argentina (Patagonia) in 1996, a new mechanism of hantavirus spread was described for Andes virus (AND): person-to-person transmission. Since then, this mode of transmission has been limited to the endemic area of AND Sout lineage. … We report here an HPS case, patient A, with residence in Buenos Aires Province, whose unique epidemiological risk factor was to have shared a trip to Southern Argentina with an already ill person, later confirmed as an HPS case (patient I). A third case (patient B), with residence in Buenos Aires Province, developed HPS after several contact events with patient A, with no additional risk factors. … Hantavirus infection was confirmed in all three patients by serological diagnosis and viral genetic characterization. The viral lineage characterized was AND Sout, being the same strain in the three cases. The clinical picture of patient A showed profound compromise, with gastrointestinal symptoms, with no clinical evidence of pulmonary involvement. … In this work we present new evidence supporting person-to-person transmission for AND Sout lineage, where there was an only contact with the index case, occasional, prolonged and non-repeated, established out of the endemic area for AND Sout lineage.
-
- Valeria P Martínez et al. 2020-12. “Super-Spreaders” and Person-to-Person Transmission of Andes Virus in Argentina”. N Engl J Med. 2020 Dec 3;383(23):2230-2241. doi: 10.1056/NEJMoa2009040. https://pubmed.ncbi.nlm.nih.gov/33264545/
- super-spreading
-
Conclusions: Among patients with ANDV hantavirus pulmonary syndrome, high viral titers in combination with attendance at massive social gatherings or extensive contact among persons were associated with a higher likelihood of transmission.
Influenza
- from 1890:

-
Whether influenza is directly contagious is not yet decided, but the author considers it probable, though the great majority of cases contract the disease by infection through the inspiring of the germs in the air
- Alford et al. 1966. Human influenza resulting from aerosol inhalation. https://journals.sagepub.com/doi/abs/10.3181/00379727-122-31255.
-
Summary. Volunteers were given A2 influenza virus in a small-particle aerosol. Infection and typical influenza resulted from low doses of virus administered in this manner. Low levels of serum neutralizing antibody were not completely effective in preventing infection and illness. The human infectious dose of this influenza strain when administered by aerosol to subjects free of serum neutralizing antibody was approximately 3 TCID50.
-
- Moser 1979. https://academic.oup.com/aje/article-abstract/110/1/1/62223?redirectedFrom=fulltext.
- influenza outbreak on a plane which strongly suggests airborne spread
- Goldmann in 2000.
- Not a detection paper, but commenting on Mozer - outbreak of flu on a plane on the ground (ventilation shut off) which took out almost everybody except the 5 ppl that got off.
- Some Canadian authors said about that outbreak “possible people wandered the plane”. Haha. What a lark.

found influenza virus in the air - see the earlier section
found virus or its genetic material in the air or in places unlikely to be touched
- Fabian 2008.
- https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0002691
- RNA out of breath sample
- Stelzer-Braid 2009. https://pubmed.ncbi.nlm.nih.gov/19626609/
- multiple viruses found in breath samples, including flu
- Blachere 2009. https://academic.oup.com/cid/article/48/4/438/283890
- Measurement of Airborne Influenza Virus in a Hospital Emergency Department
- Lindsley 2010. https://pubmed.ncbi.nlm.nih.gov/20100093/
- Yang 2011. https://pubmed.ncbi.nlm.nih.gov/21300628/
- Noti 2012. https://academic.oup.com/cid/article/54/11/1569/321689
- Bischoff 2013. https://pubmed.ncbi.nlm.nih.gov/23372182/
- Nikitin 2014. https://www.hindawi.com/journals/av/2014/859090/
- Leung 2016. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4743992/
- Yan 2018. https://www.pnas.org/content/115/5/1081
- Coleman 2018. https://pubmed.ncbi.nlm.nih.gov/30504827/
- Xie 2019. https://academic.oup.com/cid/article/70/5/850/5432328?login=true
Reports of outbreaks that occurred despite using contact precautions
- Wong 2010. https://pubmed.ncbi.nlm.nih.gov/20942655/
- spread despite use of “droplet & contact” precautions
virus remained viable in air over a significant period of time
- Wells 1936. https://pubmed.ncbi.nlm.nih.gov/17807363/
- Hemmes 1960. https://www.nature.com/articles/188430a0
- Sattar 1987. https://doi.org/10.1080/10643388709388331
- lists various times infuenza has lasted in air, including 24 hours (Table 1)
- Kormuth 2018. https://pubmed.ncbi.nlm.nih.gov/29878137/
human challenge trials or human to animal challenge trials
- Henle 1946. https://www.jimmunol.org/content/52/2/145.short
- Alford 1996. https://journals.sagepub.com/doi/abs/10.3181/00379727-122-31255
- Bischoff 2011. https://pubmed.ncbi.nlm.nih.gov/21673029/
- Nguyen van Tam 2020. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7390452/
- Bueno de Mesquita 2020. https://onlinelibrary.wiley.com/doi/epdf/10.1111/ina.12701
Kennel cough
-
Kennel cough is a syndrome from a number of viruses. The viruses

-
Here are the viruses:

aerosol and subclinical infection

- Most vaccines non-sterilizing. Note CDV for a sec.

CDV vaccine sterilizing. Let’s see how it disseminates. Ah, systematically via lymphatic system. Thus, as I said before.

Future guesses re intranasal

Finally, the opposite of what we are currently doing re SARS-CoV-2

https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7132485/
lassavirus air
https://mastodon.social/@jmcrookston/111983739817941277
lentivirus (HIV, etc) air
https://mastodon.social/@jmcrookston/111355468559961138
leprosy (bacteria) air
https://mastodon.social/@jmcrookston/110927904297332164
MRSA (bacteria)
https://mastodon.social/@jmcrookston/112170693238203259
measles
outbreak investigations which showed spread over 2 meters
- Riley 1987 https://pubmed.ncbi.nlm.nih.gov/665658/
- Bloch 1985 https://pubmed.ncbi.nlm.nih.gov/3982900/
- Remington 1985 https://pubmed.ncbi.nlm.nih.gov/3974036/
- Chen 1989
- Ehreleman 1995
norovirus
- Alsved, Malin, Anders Widell, Henrik Dahlin, Sara Karlson, Patrik Medstrand, and Jakob Löndahl. ‘Aerosolization and Recovery of Viable Murine Norovirus in an Experimental Setup’. Scientific Reports 10, no. 1 (29 September 2020): 15941. https://doi.org/10.1038/s41598-020-72932-5
rabies air
https://mastodon.social/@jmcrookston/113102340965304033
RSV and B pertussis (bacteria) air
https://mastodon.social/@jmcrookston/111466006836615698
rhinovirus air
https://mastodon.social/@jmcrookston/111467417640108406
tuberculosis
pathogen remained viable in air over a significant period of time
outbreak investigations which showed spread over 2 meters
- Houk 1980. https://pubmed.ncbi.nlm.nih.gov/6939378/
- Kenyon 1996. https://pubmed.ncbi.nlm.nih.gov/8596593/
Reports of outbreaks that occurred despite using contact precautions
- Kantor 1988. Kantor, H. S., R. Poblete, and S. L. Pusateri. “Nosocomial Transmission of Tuberculosis from Unsuspected Disease.” The American Journal of Medicine 84, no. 5 (1988): 833–38. https://doi.org/10.1016/0002-9343(88)90060-5.
- Hutton 1990. https://pubmed.ncbi.nlm.nih.gov/2105362/
Reports of true long-range airborne spread (i.e. over large distances or atmospheric)
- Hervàs, Anna, Lluís Camarero, Isabel Reche, and Emilio O. Casamayor. “Viability and Potential for Immigration of Airborne Bacteria from Africa That Reach High Mountain Lakes in Europe.” Environmental Microbiology 11, no. 6 (2009): 1612–23. https://doi.org/10.1111/j.1462-2920.2009.01926.x.
- Ssematimba, Amos, Thomas J. Hagenaars, and Mart C. M. de Jong. “Modelling the Wind-Borne Spread of Highly Pathogenic Avian Influenza Virus between Farms.” PloS One 7, no. 2 (2012): e31114. https://doi.org/10.1371/journal.pone.0031114.
- Alonso, Carmen, Dane P. Goede, Robert B. Morrison, et al. “Evidence of Infectivity of Airborne Porcine Epidemic Diarrhea Virus and Detection of Airborne Viral RNA at Long Distances from Infected Herds.” Veterinary Research 45, no. 1 (2014): 73. https://doi.org/10.1186/s13567-014-0073-z.
- Van Leuken, J.P.G., A.N. Swart, A.H. Havelaar, A. Van Pul, W. Van der Hoek, and D. Heederik. “Atmospheric Dispersion Modelling of Bioaerosols That Are Pathogenic to Humans and Livestock – A Review to Inform Risk Assessment Studies.” Microbial Risk Analysis 1 (January 2016): 19–39. https://doi.org/10.1016/j.mran.2015.07.002.
- Reche, Isabel, Gaetano D’Orta, Natalie Mladenov, Danielle M. Winget, and Curtis A. Suttle. “Deposition Rates of Viruses and Bacteria above the Atmospheric Boundary Layer.” The ISME Journal 12, no. 4 (2018): 1154–62. https://doi.org/10.1038/s41396-017-0042-4.
- MacIntyre, Chandini Raina, Arpita Das, Xin Chen, Charitha De Silva, and Con Doolan. “Evidence of Long-Distance Aerial Convection of Variola Virus and Implications for Disease Control.” Viruses 12, no. 1 (2020): 1. https://doi.org/10.3390/v12010033.
- Nagy, Alexander, Lenka Černíková, and Kamil Sedlák. “Genetic Data and Meteorological Conditions Suggesting Windborne Transmission of H5N1 High-Pathogenicity Avian Influenza between Commercial Poultry Outbreaks.” PloS One 20, no. 9 (2025): e0319880. https://doi.org/10.1371/journal.pone.0319880.
Quantifying virus exhaled from the breath
- Alsved, M., A. Matamis, R. Bohlin, et al. “Exhaled Respiratory Particles during Singing and Talking.” Aerosol Science and Technology 54, no. 11 (2020): 1245–48. https://doi.org/10.1080/02786826.2020.1812502.
- Alsved, Malin, Kristina Nyström, Sara Thuresson, et al. “Infectivity of Exhaled SARS-CoV-2 Aerosols Is Sufficient to Transmit Covid-19 within Minutes.” Scientific Reports 13, no. 1 (2023): 1. https://doi.org/10.1038/s41598-023-47829-8.
- Anand, S., and Y. S. Mayya. “Size Distribution of Virus Laden Droplets from Expiratory Ejecta of Infected Subjects.” Scientific Reports 10, no. 1 (2020): 21174. https://doi.org/10.1038/s41598-020-78110-x.
- Buonanno, G., A. Robotto, E. Brizio, et al. “Link between SARS-CoV-2 Emissions and Airborne Concentrations: Closing the Gap in Understanding.” Journal of Hazardous Materials 428 (April 2022): 128279. https://doi.org/10.1016/j.jhazmat.2022.128279.
- Buonanno, G., L. Stabile, and L. Morawska. “Estimation of Airborne Viral Emission: Quanta Emission Rate of SARS-CoV-2 for Infection Risk Assessment.” Environment International 141 (August 2020): 105794. https://doi.org/10.1016/j.envint.2020.105794.
- Chen, Paul Z., Niklas Bobrovitz, Zahra Premji, Marion Koopmans, David N. Fisman, and Frank X. Gu. “Heterogeneity in Transmissibility and Shedding SARS-CoV-2 via Droplets and Aerosols.” Elife 10 (April 2021). https://doi.org/10.7554/eLife.65774.
- Chen, Paul Z., Marion Koopmans, David N. Fisman, and Frank X. Gu. “Understanding Why Superspreading Drives the COVID-19 Pandemic but Not the H1N1 Pandemic.” The Lancet Infectious Diseases 0, no. 0 (2021). https://doi.org/10.1016/S1473-3099(21)00406-0.
- Coleman, Kristen K., Douglas Jie Wen Tay, Kai Sen Tan, et al. “Viral Load of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) in Respiratory Aerosols Emitted by Patients With Coronavirus Disease 2019 (COVID-19) While Breathing, Talking, and Singing.” Clinical Infectious Diseases 74, no. 10 (2022): 1722–28. https://doi.org/10.1093/cid/ciab691.
- Dai, Hui, and Bin Zhao. “Association between the Infection Probability of COVID-19 and Ventilation Rates: An Update for SARS-CoV-2 Variants.” Building Simulation 16, no. 1 (2023): 3–12. https://doi.org/10.1007/s12273-022-0952-6.
- Dinkele, Ryan, Sophia Gessner, Andrea McKerry, et al. “Aerosolization of Mycobacterium Tuberculosis by Tidal Breathing.” American Journal of Respiratory and Critical Care Medicine 206, no. 2 (2022): 206–16. https://doi.org/10.1164/rccm.202110-2378OC.
- Edwards, David A., Dennis Ausiello, Jonathan Salzman, et al. “Exhaled Aerosol Increases with COVID-19 Infection, Age, and Obesity.” Proc. Natl. Acad. Sci. U. S. A. 118, no. 8 (2021). https://doi.org/10.1073/pnas.2021830118.
- Fennelly, Kevin P. “Particle Sizes of Infectious Aerosols: Implications for Infection Control.” The Lancet Respiratory Medicine 8, no. 9 (2020): 914–24. https://doi.org/10.1016/S2213-2600(20)30323-4.
- Foat, Timothy G., Benjamin Higgins, Charlotte Abbs, et al. “Modeling the Effect of Temperature and Relative Humidity on Exposure to SARS-CoV-2 in a Mechanically Ventilated Room.” Indoor Air 32, no. 11 (2022): e13146. https://doi.org/10.1111/ina.13146.
- Gregson, Florence K. A., Natalie A. Watson, Christopher M. Orton, et al. “Comparing Aerosol Concentrations and Particle Size Distributions Generated by Singing, Speaking and Breathing.” Aerosol Science and Technology 55, no. 6 (2021): 681–91. https://doi.org/10.1080/02786826.2021.1883544.
- Gutmann, Desireé, Gerhard Scheuch, Timon Lehmkühler, et al. “Aerosol Measurement Identifies SARS-CoV 2 PCR Positive Adults Compared with Healthy Controls.” Environmental Research 216 (January 2023): 114417. https://doi.org/10.1016/j.envres.2022.114417.
- Hawks, Seth A., Aaron J. Prussin, Sarah C. Kuchinsky, Jin Pan, Linsey C. Marr, and Nisha K. Duggal. “Infectious SARS-CoV-2 Is Emitted in Aerosol Particles.” mBio 12, no. 5 (2021): e02527-21. https://doi.org/10.1128/mBio.02527-21.
- Hawks, Seth A., Aaron J. Prussin, Sarah C. Kuchinsky, Jin Pan, Linsey C. Marr, and Nisha K. Duggal. “Infectious SARS-CoV-2 Is Emitted in Aerosols.” Preprint, August 10, 2021. https://doi.org/10.1101/2021.08.10.455702.
- Holmgren, Helene, Evert Ljungström, Ann-Charlotte Almstrand, Björn Bake, and Anna-Carin Olin. “Size Distribution of Exhaled Particles in the Range from 0.01 to 2.0μm.” Journal of Aerosol Science 41, no. 5 (2010): 439–46. https://doi.org/10.1016/j.jaerosci.2010.02.011.
- Iqbal, Saima S. Here’s How Much Coronavirus People Infected with COVID-19 May Exhale. Health & Medicine. September 25, 2023. https://www.sciencenews.org/article/coronavirus-infected-covid-19-exhale.
- Kakeshpour, Tayeb, and Adriaan Bax. “Resolving the Loss of Intermediate-Size Speech Aerosols in Funnel-Guided Particle Counting Measurements.” Atmosphere 15, no. 5 (2024): 5. https://doi.org/10.3390/atmos15050570.
- Lane, Gregory, Guangyu Zhou, Judd F. Hultquist, Lacy M. Simons, Ramon Lorenzo-Redondo, et al. “Quantity of SARS-CoV-2 RNA Copies Exhaled per Minute during Natural Breathing over the Course of COVID-19 Infection.” Preprint, medRxiv, September 8, 2023. https://doi.org/10.1101/2023.09.06.23295138.
- Lane, Gregory, Guangyu Zhou, Judd F. Hultquist, Lacy M. Simons, Ramon Lorenzo- Redondo, et al. “Quantity of SARS-CoV-2 RNA Copies Exhaled per Minute during Natural Breathing over the Course of COVID-19 Infection.” Preprint, medRxiv, September 8, 2023. https://doi.org/10.1101/2023.09.06.23295138.
- Loudon, Robert G., and Rena Marie Roberts. “Droplet Expulsion from the Respiratory TRACT.” American Review of Respiratory Disease 95, no. 3 (1967): 435–42. https://doi.org/10.1164/arrd.1967.95.3.435.
- Ma, Jianxin, Xiao Qi, Haoxuan Chen, et al. “Exhaled Breath Is a Significant Source of SARS-CoV-2 Emission.” Preprint, medRxiv, June 2, 2020. https://doi.org/10.1101/2020.05.31.20115154.
- Malik, Madiha, Ann-Cathrin Kunze, Thomas Bahmer, Stefan Herget-Rosenthal, and Thomas Kunze. “SARS-CoV-2: Viral Loads of Exhaled Breath and Oronasopharyngeal Specimens in Hospitalized Patients with COVID-19.” International Journal of Infectious Diseases 110 (September 2021): 105–10. https://doi.org/10.1016/j.ijid.2021.07.012.
- Morawska, L., G. R. Johnson, Z. D. Ristovski, et al. “Size Distribution and Sites of Origin of Droplets Expelled from the Human Respiratory Tract during Expiratory Activities.” Journal of Aerosol Science 40, no. 3 (2009): 256–69. https://doi.org/10.1016/j.jaerosci.2008.11.002.
- Mürbe, Dirk, Martin Kriegel, Julia Lange, Lukas Schumann, Anne Hartmann, and Mario Fleischer. “Aerosol Emission of Adolescents Voices during Speaking, Singing and Shouting.” PLOS ONE 16 (February 2021): e0246819. https://doi.org/10.1371/journal.pone.0246819.
- Nishandar, Sanika Ravindra, Yucheng He, Marko Princevac, and Rufus D. Edwards. “Fate of Exhaled Droplets From Breathing and Coughing in Supermarket Checkouts and Passenger Cars.” Environmental Health Insights, ahead of print, January 10, 2023. Sage UK: London, England. https://doi.org/10.1177/11786302221148274.
- Papineni, Rao S., and Frank S. Rosenthal. “The Size Distribution of Droplets in the Exhaled Breath of Healthy Human Subjects.” Journal of Aerosol Medicine 10, no. 2 (1997): 105–16. https://doi.org/10.1089/jam.1997.10.105.
- Parhizkar, Hooman, Leslie Dietz, Andreas Olsen-Martinez, et al. “Quantifying Environmental Mitigation of Aerosol Viral Load in a Controlled Chamber With Participants Diagnosed With Coronavirus Disease 2019.” Clinical Infectious Diseases 75, no. 1 (2022): e174–84. https://doi.org/10.1093/cid/ciac006.
- Parhizkar, Hooman, Leslie Dietz, Andreas Olsen-Martinez, et al. “Quantifying Human and Environmental Viral Load Relationships amidst Mitigation Strategies in a Controlled Chamber with Participants Having COVID-19.” Preprint, October 1, 2021. https://doi.org/10.21203/rs.3.rs-940891/v1.
- Pöhlker, Mira L., Ovid O. Krüger, Jan-David Förster, et al. “Respiratory Aerosols and Droplets in the Transmission of Infectious Diseases.” arXiv:2103.01188. Preprint, arXiv, August 4, 2021. http://arxiv.org/abs/2103.01188.
- Pöhlker, Mira L., Christopher Pöhlker, Ovid O. Krüger, et al. “Respiratory Aerosols and Droplets in the Transmission of Infectious Diseases.” Reviews of Modern Physics 95, no. 4 (2023): 045001. https://doi.org/10.1103/RevModPhys.95.045001.
- Riediker, Michael, and Dai-Hua Tsai. “Estimation of Viral Aerosol Emissions From Simulated Individuals With Asymptomatic to Moderate Coronavirus Disease 2019.” JAMA Network Open 3, no. 7 (2020): e2013807. https://doi.org/10.1001/jamanetworkopen.2020.13807.
- Shen, Yang, Joseph M. Courtney, Philip Anfinrud, and Adriaan Bax. “Hybrid Measurement of Respiratory Aerosol Reveals a Dominant Coarse Fraction Resulting from Speech That Remains Airborne for Minutes.” Proceedings of the National Academy of Sciences 119, no. 26 (2022): e2203086119. https://doi.org/10.1073/pnas.2203086119.
- Tang, Julian W., Andre D. Nicolle, Christian A. Klettner, et al. “Airflow Dynamics of Human Jets: Sneezing and Breathing - Potential Sources of Infectious Aerosols.” PLOS ONE 8, no. 4 (2013): e59970. https://doi.org/10.1371/journal.pone.0059970.
- Trivedi, Shrey, Savvas Gkantonas, Léo C. C. Mesquita, Salvatore Iavarone, Pedro M. de Oliveira, and Epaminondas Mastorakos. “Estimates of the Stochasticity of Droplet Dispersion by a Cough.” Physics of Fluids 33, no. 11 (2021): 115130. https://doi.org/10.1063/5.0070528.
- Vernez, David, Sophie Schwarz, Jean-Jacques Sauvain, Christiane Petignat, and Guillaume Suarez. “Probable Aerosol Transmission of SARS-CoV-2 in a Poorly Ventilated Courtroom.” Indoor Air, ahead of print, June 11, 2021. https://doi.org/10.1111/ina.12866.
- Wurie, Fatima, Olivier Le Polain de Waroux, Matthew Brande, et al. “Characteristics of Exhaled Particle Production in Healthy Volunteers: Possible Implications for Infectious Disease Transmission.” 2:14. Preprint, F1000Research, January 15, 2013. https://doi.org/10.12688/f1000research.2-14.v1.
- Zhou, Jie, Anika Singanayagam, Niluka Goonawardane, et al. “Viral Emissions into the Air and Environment after SARS-CoV-2 Human Challenge: A Phase 1, Open Label, First-in-Human Study.” The Lancet Microbe 0, no. 0 (2023). https://doi.org/10.1016/S2666-5247(23)00101-5.
- Zhou, Jie, Jianjian Wei, Ka-Tim Choy, et al. “Defining the Sizes of Airborne Particles That Mediate Influenza Transmission in Ferrets.” Proceedings of the National Academy of Sciences of the United States of America 115, no. 10 (2018): E2386–92. https://doi.org/10.1073/pnas.1716771115.
Miscellaneous other topics
There isn’t a single study proving “droplet” transmission
See https://x.com/jmcrookston/status/1415368175592562696#m.
There is little support for fomites
A bunch of fomite articles just to see how they described fomite spread. Turns out, pretty weakly. See https://x.com/jmcrookston/status/1334851435444531200#m.
Review articles about coronaviruses
A thread with review articles about coronaviruses. They are a great place to learn all that we already knew about coronaviruses before this pandemic started. You know, from our 50 years of dealing with them. Tl;dr: most of what people “discovered” about CoVs, we already knew. Including kids, not sterilizing, persistence, in brain, etc.
https://x.com/jmcrookston/status/1310275748108922881#m
hantavirus, articles either about it transmitting person-to-person or by airborne
- Enría, D., P. Padula, E. L. Segura, et al. “Hantavirus Pulmonary Syndrome in Argentina. Possibility of Person to Person Transmission.” Medicina 56, no. 6 (1996): 709–11.
- Wells, R. M., S. Sosa Estani, Z. E. Yadon, et al. “An Unusual Hantavirus Outbreak in Southern Argentina: Person-to-Person Transmission? Hantavirus Pulmonary Syndrome Study Group for Patagonia.” Emerging Infectious Diseases 3, no. 2 (1997): 171–74. https://doi.org/10.3201/eid0302.970210.
- Macready, Norra. “Hantavirus May Spread from Person to Person.” The Lancet 349, no. 9069 (1997): 1890. https://doi.org/10.1016/S0140-6736(05)63882-2.
- Markotic, Alemka. “Human-to-Human Transmission of Hantaviruses.” The Lancet 350, no. 9077 (1997): 596. https://doi.org/10.1016/S0140-6736(05)63191-1.
- Castillo, C., J. Mardones, and E. Villagra. “[Prevalence of anti-hantavirus antibodies in health care personnel in direct contact with patients with hantavirus pulmonary syndrome in Temuco, Chile 1997 to 1999].” Revista Medica De Chile 128, no. 7 (2000): 735–39.
- Castillo, Constanza, Eliecer Villagra, Ligia Sanhueza, Marcela Ferres, Jovita Mardones, and Gregory J. Mertz. “Prevalence of Antibodies to Hantavirus among Family and Health Care Worker Contacts of Persons with Hantavirus Cardiopulmonary Syndrome: Lack of Evidence for Nosocomial Transmission of Andes Virus to Health Care Workers in Chile.” The American Journal of Tropical Medicine and Hygiene 70, no. 3 (2004): 302–4.
- Pinna, Diego M., Valeria P. Martínez, Carla M. Bellomo, Claudia López, and Paula Padula. “[New epidemiologic and molecular evidence of person to person transmission of hantavirus Andes Sout].” Medicina 64, no. 1 (2004): 43–46.
- 2005-12. Valeria P Martinez, Carla Bellomo, Jorge San Juan, Diego Pinna, Raul Forlenza, Malco Elder, Paula J Padula. 2005. Person-to-person transmission of Andes virus. Emerg Infect Dis. 2005 Dec;11(12):1848-53. doi: 10.3201/eid1112.050501. https://pubmed.ncbi.nlm.nih.gov/16485469/
-
Despite the fact that rodents are considered to be the infectious source of hantavirus for humans, another route of transmission was demonstrated. Andes virus (ANDV) has been responsible for most of the cases recorded in Argentina. Person-to-person transmission of ANDV Sout lineage was described during an outbreak of hantavirus pulmonary syndrome in southwest Argentina. In this study, we analyzed 4 clusters that occurred in 2 disease-endemic areas for different ANDV lineages. We found new evidence of interhuman transmission for ANDV Sout lineage and described the first event in which another lineage, ANDV Cent BsAs, was implicated in this mechanism of transmission. On the basis of epidemiologic and genetic data, we concluded that person-to-person spread of the virus likely took place during the prodromal phase or shortly after it ended, since close and prolonged contact occurred in the events analyzed here, and the incubation period was 15-24 days.
-
Despite the fact that rodents are considered to be the infectious source of hantavirus for humans, another route of transmission was demonstrated. Andes virus (ANDV) has been responsible for most of the cases recorded in Argentina. Person-to-person transmission of ANDV Sout lineage was described during an outbreak of hantavirus pulmonary syndrome in southwest Argentina. In this study, we analyzed 4 clusters that occurred in 2 disease-endemic areas for different ANDV lineages. We found new evidence of interhuman transmission for ANDV Sout lineage and described the first event in which another lineage, ANDV Cent BsAs, was implicated in this mechanism of transmission.
-
- 2014-10. Constanza Martinez-Valdebenito, Mario Calvo, Cecilia Vial, Rita Mansilla, Claudia Marco, R Eduardo Palma, Pablo A Vial, Francisca Valdivieso, Gregory Mertz, Marcela Ferrés. 2011. “Person-to-person household and nosocomial transmission of andes hantavirus, Southern Chile 2011” PMID: 25272189 PMCID: PMC4193174 DOI: 10.3201/eid2010.140353. https://pubmed.ncbi.nlm.nih.gov/25272189/
-
Abstract. Andes hantavirus (ANDV) causes hantavirus cardiopulmonary syndrome in Chile and is the only hantavirus for which person-to-person transmission has been proven. We describe an outbreak of 5 human cases of ANDV infection in which symptoms developed in 2 household contacts and 2 health care workers after exposure to the index case-patient. Results of an epidemiologic investigation and sequence analysis of the virus isolates support person-to-person transmission of ANDV for the 4 secondary case-patients, including nosocomial transmission for the 2 health care workers. Health care personnel who have direct contact with ANDV case-patients or their body fluids should take precautions to prevent transmission of the virus. In addition, because the incubation period of ANDV after environmental exposure is longer than that for person-to-person exposure, all persons exposed to a confirmed ANDV case-patient or with possible environmental exposure to the virus should be monitored for 42 days for clinical symptoms.
-
…Results of an epidemiologic investigation and sequence analysis of the virus isolates support person-to-person transmission of ANDV for the 4 secondary case-patients, including nosocomial transmission for the 2 health care workers.”
-
“We describe an outbreak of 5 human cases of ANDV infection in which symptoms developed in 2 household contacts and 2 health care workers after exposure to the index case-patient.” - Martinez-Valdebenito et al., 2014, p. 1629
-
“Although person-to-person transmission in Chile has been epidemiologically documented (10), nosocomial transmission has not been reported.”- Martinez-Valdebenito et al., 2014, p. 1629
-
“Case-patient C was the spouse of case-patient A. She shared the same bed and cared for him during his febrile prodrome but denied that they had sexual activity after symptom onset.” - Martinez-Valdebenito et al., 2014, p. 1632
-
“On March 18, twenty-five days after her husband’s illness onset and 41 days after they entered the cellar, she exhibited mild fever, severe headache, myalgia, and photophobia .” - Martinez-Valdebenito et al., 2014, p. 1632
-
“Case-patient D, a housekeeper at hospital I, had fever, abdominal pain, and vomiting develop on March 18. Two days later, she was hospitalized at hospital I, and 4 days later, she was transferred to hospital II, where severe shock and respiratory failure developed. She died a few hours after admission to hospital II.” - Martinez-Valdebenito et al., 2014, p. 1632.
-
“First, the period of the disease during which the acute case-patient and the household contact or health care personnel have close contact is primarily the febrile prodrome phase, when symptoms are nonspecific for hantavirus” - Martinez-Valdebenito et al., 2014, p. 1633
-
“Second, the number of days from exposure to an index case-patient and the onset of symptoms among additional cases ranges from 12 to 27 days (7,26), consistent with the intervals observed in our report.” - Martinez-Valdebenito et al., 2014, p. 1633
-
“One case of nosocomial transmission of the virus has been previously reported in Argentina (7), and evidence of this transmission has been sought in Chile (12). We document 2 cases of nosocomial transmission of ANDV, from the index case-patient to a nursing assistant and to a housekeeper, even though their contact with the patient was limited to kissing the patient on the cheek and to” - Martinez-Valdebenito et al., 2014, p. 1633
-
“handling bedding and gowns (no invasive procedures).” - Martinez-Valdebenito et al., 2014, p. 1634
-
“Our study documents a small but definite risk of nosocomial acquisition of ANDV infection for personnel who care for patients, including handling of bedding and gowns. After this investigation, the Ministry of Health of Chile has recommended, in addition to strict adherence to universal precautions, the use of droplet precautions when ANDV infection is suspected. Use of N95 respirator masks, designed to prevent the inhalation of airborne particles, is recommended for those procedures associated with aerosolization of viral contaminated secretions (e.g., respiratory, saliva) when procedures such as suction or intubation are performed. However, this recommendation should be extended to all personnel who have any kind of direct contact with patients or body fluids, including bedding and gowns.” - Martinez-Valdebenito et al., 2014, p. 1634.
-
- Pettersson, Lisa, Jonas Klingström, Jonas Hardestam, Ake Lundkvist, Clas Ahlm, and Magnus Evander. “Hantavirus RNA in Saliva from Patients with Hemorrhagic Fever with Renal Syndrome.” Emerging Infectious Diseases 14, no. 3 (2008): 406–11. https://doi.org/10.3201/eid1403.071242.
- Martínez, Valeria P., Nicholas Di Paola, Daniel O. Alonso, et al. “‘Super-Spreaders’ and Person-to-Person Transmission of Andes Virus in Argentina.” The New England Journal of Medicine 383, no. 23 (2020): 2230–41. https://doi.org/10.1056/NEJMoa2009040.